
ORG.CHEM.W/LSLF.SOL.MAN. >P<
9th Edition
ISBN: 9780134581040
Author: Wade
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.53SP
Each of these compounds can react as a nucleophile In each case use curved arrows to show how the nucleophile would react with the strong electrophile BF3.
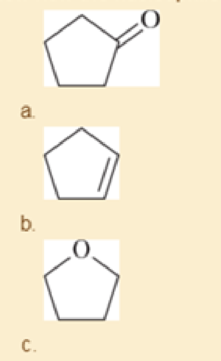
- d. (CH3)3N
- e. CH3CH2OH
- f. (CH3)2S
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the species in each group in order of increasing nucleophilicity.
a. CH3CH2S−CH3CH2O−, CH3CO2− in CH3OH
b. CH3NH2, CH3SH, CHOH in acetone
c.−OH, F−, Cl− in acetone
d. HS−, F−, Cl− in CH3OH
A
3. Rank the following compounds in terms of reactivity with a good nucleophile.
Br
B
NH₂
F
D
OH
E
CI
Write out the expected splitting pattern for hydrogens Ha, Hp and Hc
3Jab = 10 Hz
На
Hb
3Jpc = 7 Hz
CI
Hc
Hc
Ha:Hb:Hc: doublet, quartet, doublet
Ha:Hb:Hc: quartet, quartet, triplet
Ha:Hb:Hc: doublet, doublet of triplets, doublet
Ha:Hb:Hc: doublet, triplet of doublets, doublet
Chapter 2 Solutions
ORG.CHEM.W/LSLF.SOL.MAN. >P<
Ch. 2.1A - Prob. 2.1PCh. 2.1B - The NF bond is more polar than the NH bond: but...Ch. 2.1B - For each of the following compounds 1. Draw the...Ch. 2.1B - Two isomers of 1,2-dichloroethene are known One...Ch. 2.2C - Prob. 2.5PCh. 2.2C - Prob. 2.6PCh. 2.3 - Prob. 2.7PCh. 2.4 - Calculate the pH of the following solutions a....Ch. 2.6A - Ammonia appears in Table 2-2 as both an acid and a...Ch. 2.7 - Write equations for the following acid-base...
Ch. 2.7 - Ethanol, methylamine. and acetic acid are all...Ch. 2.8 - Prob. 2.12PCh. 2.10 - Write equations for the following acid-base...Ch. 2.10 - Rank the following acids in decreasing order of...Ch. 2.11 - Prob. 2.15PCh. 2.11 - Prob. 2.16PCh. 2.11 - Consider each pair of bases and explain which one...Ch. 2.12 - Which is a stronger base ethoxide ion or acetate...Ch. 2.12 - Prob. 2.19PCh. 2.12 - Prob. 2.20PCh. 2.12 - Prob. 2.21PCh. 2.12 - Choose the more basic member of each pair of...Ch. 2.14 - Prob. 2.23PCh. 2.15D - Classify the following hydrocarbons and draw a...Ch. 2.16D - Prob. 2.25PCh. 2.17C - Draw a Lewis structure and classify each of the...Ch. 2.17C - Circle the functional groups in the following...Ch. 2 - The CN triple bond in acetonitrile has a dipole...Ch. 2 - Prob. 2.29SPCh. 2 - Sulfur dioxide has a dipole moment of 1.60 D....Ch. 2 - Which of the following pure compounds can form...Ch. 2 - Predict which member of each pair is more soluble...Ch. 2 - Prob. 2.33SPCh. 2 - Prob. 2.34SPCh. 2 - Predict which compound in each pair has the higher...Ch. 2 - All of the following compounds can react as acids...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - The Ka of phenylacetic acid is 5 2 105, and the...Ch. 2 - The following compound can become protonated on...Ch. 2 - The following compounds are listed in increasing...Ch. 2 - Prob. 2.42SPCh. 2 - Prob. 2.43SPCh. 2 - Compare the relative acidity of 1-molar aqueous...Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - The following compounds can all react as bases. a....Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - Prob. 2.48SPCh. 2 - Methyllithium (CH3Li) is often used as a base in...Ch. 2 - Label the reactants in these acid-base reactions...Ch. 2 - In each reaction, label the reactants as Lewis...Ch. 2 - Prob. 2.52SPCh. 2 - Each of these compounds can react as a nucleophile...Ch. 2 - Prob. 2.54SPCh. 2 - Give a definition and an example for each class of...Ch. 2 - Circle the functional groups in the following...Ch. 2 - Prob. 2.57SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In the following reaction which species acts as the nucleophile? OCH CH3 (H.C).NOCH CH CH,CH OH OCH,CH , OCH,CH, A. OCH,CH, (H,C),N B. C. CH,CH,OH D. (D)arrow_forwardThese reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4arrow_forwardQ4. The herbicide oxyfluorfen can be prepared by the reaction between phenol and an aryl fluoride. Propose a curved arrow mechanism for this reaction. 1 F3C. LOCH2CH3 F3C. КОН CI `NO2 CI OH LOCH2CH3 `NO2 Oxyfluorfenarrow_forward
- How does gold help to catalyze the reaction in the gold catalysis experiment? It deprotonates the methanol, making methanol a better nucleophile. оа. o b. It donates electron density to the alkyne, making the alkyne a better nucleophile. О с. It coordinates to the methanol, making methanol a better nucleophile. o d. It coordinates to the alkyne, making the alkyne a better electrophile.arrow_forwardWhich of the following molecules is most nucleophilic? OA. NaOCH₂CH3 OB. CH3CH₂OH OC. NaOC6H5 OD. C₂H5OH 1 E.CH3COONaarrow_forwardWhat is the product of the reaction of bromoethane with each of the following nucleophiles? a. CH3CH2CH2O− b. CH3C‚C- c. (CH3)3N d. CH3CH2S-arrow_forward
- Are the following nucleophiles, electrophiles, they be both a nucleophile or electrophile, or neither? pyridine alkenes Hg+ (ex. Hg(OAc)2 CH3ONa PBr3arrow_forward6. The following reaction is an example of OH A) SN1 B) SN2 Br NaOH EtOH C) E1 D) E2 7. In the above reaction (Q6), what is the role of NaOH? A) It acts as an acid and reacts with EtOH B) It acts as nucleophile and attacks on electrophilic carbon in bromoprop C) It acts as a base and reacts with phenolic OH and forms phenolate ion D) It acts as a spectatorarrow_forwarde) sec-butyl bromide • Question 2: Identify the nucleophile and leaving group and draw the products of each reaction. a. b. Br + + OCH₂CH3 NaOH d. Br + N3 + NaCNarrow_forward
- Ql- Arrange the following carbonium ion as more stable. Why? Me Me „Me A C Q2- Choose the substituted group that makes the following reaction faster. Why? X- CH2 H X- ососн, OR COOCH} Y= NH2 OR NO2 Q3- Which of the following compounds give the highest yield when reacting with a carbonyl compound. Why? NH 2 NH2 NH2 Me Me Me OR OR NO 2 NO2 NO2arrow_forwardIdentify the stronger nucleophile in each pair. a.NH3, −NH2 b.CH3NH2, CH3OH c.CH3CO2−, CH3CH2O−arrow_forwardSynthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates. COOH c. CH3- -CH,NH2 е. Но- -CH3 a. Br OH b. d. CH;CH2- -COOH -N=N- -NH2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License