
Interpretation:
To explain the difference in the observed pK values for P and Q.
Concept introduction:
Acid base reaction or Proton transfer reaction. It is the equilibrium reaction. An acid donates a proton to a based and thus forming conjugate acid and base. The reaction is always involves two acid and bases. It is equilibrium reaction so the position of the equilibrium depends on the strength of the acid and bases. The stronger is the acid that donates proton and the stronger base accept the proton. Then these two forms the conjugate acid and base as shown below in the Fig.
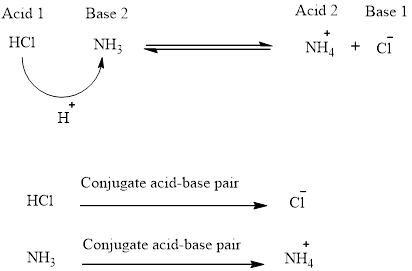
From the Bronsted-Lowry, an acid is a proton donor and from the Lewis electron pairs acceptor. Thus an acid strength is defined as the tendency to donate proton. The decision of an acid is stronger is based on the compound donating proton. If the compound readily donates the proton then it is strong acid. The strong acid is determined using the acidity constant, Ka. So larger the Ka value stronger the acid. Moreover for exponent value it is conveniently measured by pKa values.
pKa = -log Ka
So, smaller the pKa, stronger is the acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
ORGANIC CHEMISTRY-STUDY GDE...-W/ACCESS
- Explain the differences between semi-empirical and ab initio methods of electronic structure computation.arrow_forwardThank you, but why you used the 0.0010 M (mol/dm^3) when calculating the Kb without converting it to mol/m^3 so we can have a coherent units?arrow_forwardWhat states of (i) benzene, (ii) naphthalene may be reached by electric dipole transitions from their (totally symmetrical) ground states?arrow_forward
- Please answer subparts iv, v and viarrow_forwardConstruct the expression for Ksp for solid Ba₃(PO₄)₂ in aqueous solution. Ba₃(PO₄)₂(s) ⇌ 3 Ba²⁺(aq) + 2 PO₄³⁻(aq)arrow_forwardIs a method better if the LOD (limit of detection) and LLOQ (lower limit of quantitation) are of lower concentrations? Why or why not? Explain in detail.arrow_forward
- Which of the normal modes of (i) H2O, (ii) H2CO are Raman active?arrow_forwardDetermine the possible values for s2p1 configuration and use them to prepare a microstate table and assign the ground state term.arrow_forwardWhat resolution is required to seperate two lines at wavelength1 and wavelength2?arrow_forward
- What is the most highly populated rotational level of Br2 at (i) 25 °C, (ii) 100 °C? Take ᷉ B = 0.0809 cm−1.arrow_forwardPredict the intensity distribution in the hyperfine lines of the EPR spectra of (a) .CH3, (b) .CD3.arrow_forwardUse a group theoretical argument to decide which of the following transitions are electric-dipole allowed: (a) the π* ← π transition in ethene, (b) the π* ← n transition in a carbonyl group in a C2v environment.arrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning



