
Concept explainers
The Dieterici equation of state for one mole of gas is
Where a and b are constants determined experimentally. For NH3(g), a = 10.91 atm. L2 and b = 0.0401 L. Plot the pressure of the gas as the volume of 1.00 mol of NH3(g) expands from 22.4 L to 50.0 L at 273 K, and numerically determine the work done by the gas by measuring the area under the curve.
Interpretation:
The Dieterici equation of state for one mole of gas is
Where a and b are constants determined experimentally. For NH3(g), a = 10.91 atm. L2 and b = 0.0401 L. The pressure of the gas as the volume of 1.00 mol of NH3(g) expands from 22.4 L to 50.0 L at 273 K is to be plotted and numerically the work done by the gas by measuring the area under the curve is to be determined.
Concept introduction:
The ideal gas law considered the molecules of a gas as point particles with perfectly elastic collisions among them in nature. This works importantly well for gases at dilution and at low pressure in many experimental calculations. But the gas molecules are not performing as point masses, and there are situations where the properties of the gas molecules have measurable effect by experiments. Thus, a modification of the ideal gas equation was coined by Johannes D. van der Waals in 1873 to consider size of molecules and the interaction forces among them. Berthelot modified the van der Waals equation as modified Berthelot model of state and further changes was made, and the equation was provided as Dieterici equation of state. The significant advantages of this equation, such as a more realistic critical compressibility factor are documented.
Answer to Problem 2.91E
The Dieterici equation of state for one mole of gas is
Explanation of Solution
The Dieterici equation of state for one mole of gas is
Given,
a = 10.91 atm. L2
b = 0.0401 L
volume of gas initial = 22.4 L
volume of gas final = 50.0 L
temperature of system = 273 K
pressure at 22.4 L is calculated as,
∴ pressure at 22.4 L = 0.9794 atm
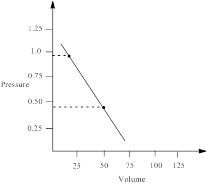
Similarly, pressure at 50 L is calculated as follows;
∴ pressure at 50 L = 0.4435 atm
From the graph the pressure difference can be calculated as;
0.5359 atm.
the work done by the gas by measuring the area under the curve is determined as;
Thus, the pressure of the gas is plotted against volume and the work done in expansion is calculated.
Want to see more full solutions like this?
Chapter 2 Solutions
Physical Chemistry
- 5-107 If 60.0 g of NH3 occupies 35.1 L under a pressure of 77.2 in. Hg, what is the temperature of the gas, in °C?arrow_forwardWhat is the value of FP for a sample of gas whose temperature is -33.0 C and volume is 0.0250 L? What temperature is required to change the volume to 66.9 cm3?arrow_forwardA cylinder of compressed gas is labeled Composition (mole %): 4.5% H2S, 3.0% CO2, balance N2. The pressure gauge attached to the cylinder reads 46 atm. Calculate the partial pressure of each gas, in atmospheres, in the cylinder.arrow_forward
- At the critical point for carbon dioxide, the substance is very far from being an ideal gas. Prove this statement by calculating the density of an ideal gas in g/cm3 at the conditions of the critical point and comparing it with the experimental value. Compute the experimental value from the fact that a mole of CO2 at its critical point occupies 94 cm3.arrow_forwardperform stoichiometric ca1cu1uions for reactions involving gases as reactants or products.arrow_forwardYou have a gas, one of the three known phosphorus-fluorine compounds (PF3, PF3, and P2F4). To find out which, you have decided to measure its molar mass. (a) First, yon determine that the density of the gas is 5.60 g/L at a pressure of 0.971 atm and a temperature of 18.2 C. Calculate the molar mass and identify the compound. (b) To check the results from part (a), you decide to measure the molar mass based on the relative rales of effusion of the unknown gas and CO2. You find that CO2 effuses at a rate of 0.050 mol/min, whereas the unknown phosphorus fluoride effuses at a rate of 0.028 mol/min. Calculate the molar mass of the unknown gas based on these results.arrow_forward
- Given that a sample of air is made up of nitrogen, oxygen, and argon in the mole fractions 0.78 N2, 0.21 O2, and 0.010 Ar, what is the density of air at standard temperature and pressure?arrow_forwardA 1.000-g sample of an unknown gas at 0C gives the following data: P(atm) V (L) 0.2500 3.1908 0.5000 1.5928 0.7500 1.0601 1.0000 0.7930 Use these data to calculate the value of the molar mass at each of the given pressures from the ideal gas law (we will call this the apparent molar mass at this pressure). Plot the apparent molar masses against pressure and extrapolate to find the molar mass at zero pressure. Because the ideal gas law is most accurate at low pressures, this extrapolation will give an accurate value for the molar mass. What is the accurate molar mass?arrow_forward93 The complete combustion of octane can be used as a model for the burning of gasoline: 2C8H18+25O216CO2+18H2O Assuming that this equation provides a reasonable model of the actual combustion process, what volume of air at 1.0 atm and 25°C must be taken into an engine to burn 1 gallon of gasoline? (The partial pressure of oxygen in air is 0.21 atm and the density of liquid octane is 0.70 g/mL.)arrow_forward
- Urea (H2NCONH2) is used extensively as a nitrogen source in fertilizers. It is produced commercially from the reaction of ammonia and carbon dioxide: 2NH3(g)+CO2(g)PressureHeatH2NCONH2(s)+H2O(g) Ammonia gas at 223C and 90. atm flows into a reactor at a rate of 500. L/min. Carbon dioxide at 223C and 45 atm flows into the reactor at a rate of 600. L/min. What mass of urea is produced per minute by this reaction assuming 100% yield?arrow_forwardYou have an equimolar mixture of the gases SO2 and O2, along with some He, in a container fitted with a piston. The density of this mixture at STP is 1.924 g/L. Assume ideal behavior and constant temperature and pressure. a. What is the mole fraction of He in the original mixture? b. The SO2 and O2 react to completion to form SO3. What is the density of the gas mixture after the reaction is complete?arrow_forwardThe Berthelot equation of state for one mole of gas is p=RTV-b-aTV2 Where a and b are constants determined experimentally. For NH3g, a = 741.6 atm. L2. K and b = 0.0139 L. Calculate p when V=22.41L and T = 273.15K. How much does the pressure vary from p as predicted by the ideal gas law?arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





