
Concept explainers
(a)
Interpretation:
Lewis structure of formate ion
Concept Introduction:
Lewis structure represents covalent bonds and describes valence electrons configuration of atoms. The covalent bonds are depicted by lines and unshared electron pairs by pairs of dots. The sequence to write Lewis structure of some molecule is given as follows:
- The central atom is identified and various other atoms are arranged around it. This central atom so chosen is often the least electronegative.
- Total valence electrons are estimated for each atom.
- single bond is first placed between each atom pair.
- The electrons left can be allocated as unshared electron pairs or as multiple bonds around
symbol of element to satisfy the octet (or duplet) for each atom. - Add charge on the overall structure in case of polyatomic cation or anion.
(a)
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
The symbol for oxygen is
One negative charge on molecule is added up as one valence electron in the total count.
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
To complete the valence electrons of carbon, it forms a double bond with one oxygen atom.
Hence, 10 electrons are allocated as 3 lone pairs on singly bonded oxygen atom and two lone pairs on doubly bonded oxygen atom. The Lewis structure of
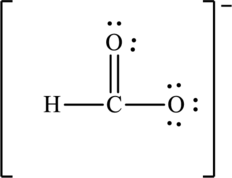
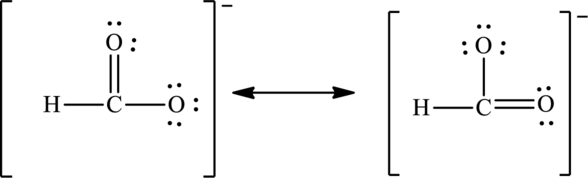
The negative charge on molecule is delocalized on each oxygen atom and since there are two oxygen atoms, therefore, two resonance structures are formed and possible resonance structures are as follows:
(b)
Interpretation:
Lewis structure of hydrogen phosphite ion
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The molecule
The symbol for oxygen is
The symbol for hydrogen is
The symbol for phosphorus is
Two negative charges on molecule are added up as two valence electrons in the total count.
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
Hence, 18 electrons are allocated as 3 lone pairs on two singly bonded oxygen atoms and 2 lone pairs on doubly bonded oxygen atom to complete their octet. The Lewis structure is as follows:
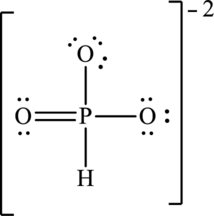
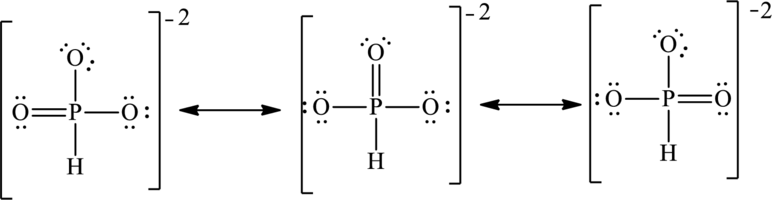
The negative charge on molecule is delocalized on three oxygen atoms therefore three resonance structures will be formed and possible resonance structures are as follows:
(c)
Interpretation:
Lewis structure of bromate ion
Concept Introduction:
Refer to part (a).
(c)
Explanation of Solution
The molecule
The symbol for oxygen is
The symbol for bromine is
One negative charge on molecule is added up to the total valence count.
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
To complete the valence electrons of bromine it forms two double bonds with two oxygen atoms.
Hence, 16 electrons are allocated as 3 lone pairs on the singly bonded oxygen atom, 2 lone pairs on doubly bonded oxygen atom and 1 lone pair on bromine to complete their respective octet. The Lewis structure is as follows:
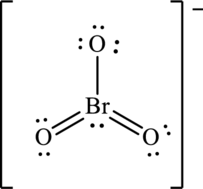
The negative charge on molecule participates in resonance by resonating on each oxygen atom. Since there are three oxygen atoms, therefore three resonating structures are drawn as:
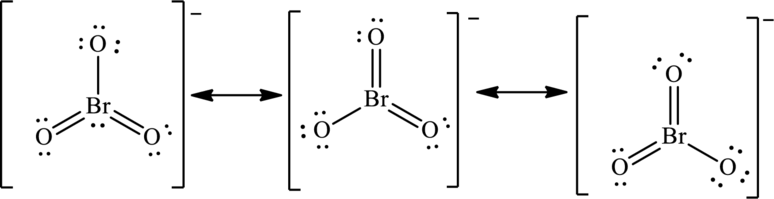
(d)
Interpretation:
Lewis structure of selenate ion
Concept Introduction:
Refer to part (a).
(d)
Explanation of Solution
The molecule
The symbol for oxygen is
The symbol for selenium is
Two negative charges on molecule are added up as two valence electrons in the total count
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
To complete the valence electrons of selenium, it forms double bond with two oxygen atom.
Hence, 20 electrons are allocated as 3 lone pairs on singly bonded oxygen atoms and 3 lone pairs on doubly bonded oxygen atoms to complete their octet. The Lewis structure is as follows:
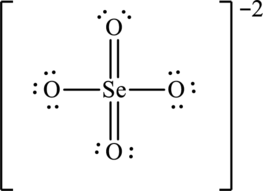
The two negative charges on molecule are delocalized on two oxygen atoms and since there are four oxygen atoms, therefore, four resonance structures will be formed and possible resonance structures are as follows:
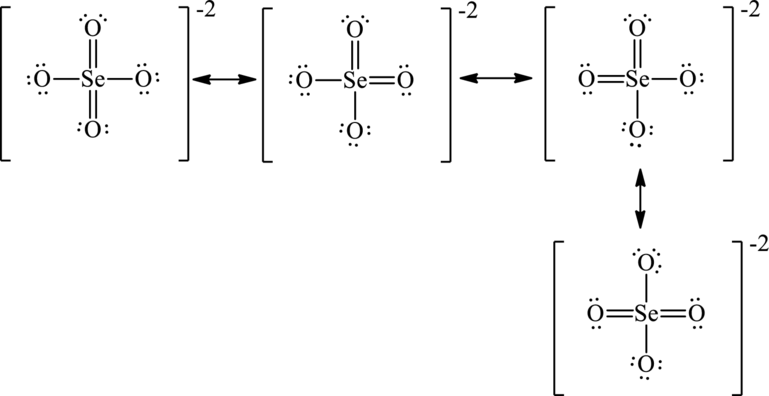
Want to see more full solutions like this?
Chapter 2 Solutions
CHEMICAL PRINCIPLES PKG W/SAPLING
- How many double bonds can be found in the best Lewis stucture of COF 2?arrow_forwardDescribe the structure of a P2 molecule in valence-bond terms. Why is P4 a more stable form of molecular phosphorus than P2?arrow_forwardDraw the Lewis structure of the azide ion, N3, and calculate the formal charge on each nitrogen atom in the structure:arrow_forward
- Several Lewis structures can be written for perbromate ion, , the central Br with all single Br—O bonds, or with one, two, or three Br=O double bonds. Draw the Lewis structures of these possible resonance structures, and use formal charges to predict which makes the greatest contribution to the resonance hybrid.arrow_forwardThe cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



