
Concept explainers
Interpretation: All the lone pairs for each of the following compounds should be drawn.
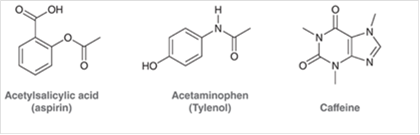
Concept Introduction The unshared pair of electrons are said to be lone pairs of electrons that are present in an atom of a compound.
Answer to Problem 34PP
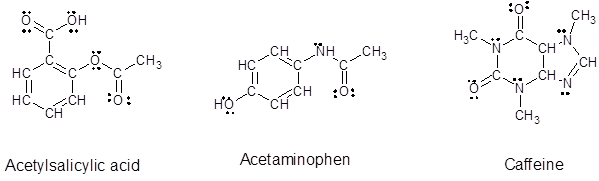
Explanation of Solution
The number of valence electrons in each isolated atom present in given compound are:
For Carbon = 4
For Hydrogen = 1
For Nitrogen = 5
For Oxygen = 6
Identify the number of bonds in the given compound:
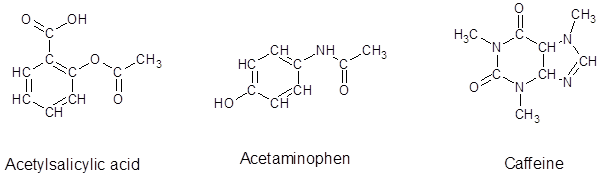
From the given structure of acetylsalicylic acid, each carbon atoms forms four bonds, hydrogen atoms forms one bond and oxygen atoms forms two bonds.
From the given structure of acetaminophen, each carbon atoms forms four bonds, hydrogen atoms forms one bond, oxygen atoms forms two bonds and nitrogen atom forms three bonds.
From the given structure of caffeine, each carbon atoms forms four bonds, hydrogen atoms forms one bond, oxygen atoms forms two bonds and nitrogen atom forms three bonds.
Since, all the valence electrons are involved in bond formation for carbon and hydrogen so there are no lone pair electrons on them. For all the oxygen atoms that forms two bonds uses two valence electrons out of six to form bond and rest four electrons are present as two lone-pair on each oxygen atom. For all the nitrogen atoms that forms three bonds uses three valence electrons out of five to form bond and rest two electrons are present as a lone-pair on each nitrogen atom. So, all the lone pairs for each compound are:
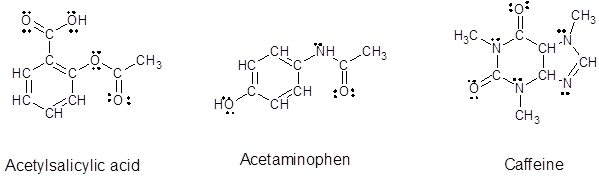
Want to see more full solutions like this?
Chapter 2 Solutions
ORGANIC CHEMISTRY 2-SEMESTER-ACCESS
- Give the name and the molecular formula (C before H) for each of the following compounds:arrow_forwardDetermine the number of lone pairs on each oxygen, nitrogen, and charged carbon atoms in the following structures.arrow_forwardThere are 17 constitutional isomers with the molecular formula of C5H13N. Draw all 17 of these isomers as skeletal structures and give the SMILES for each.arrow_forward
- Draw a skeletal structure for the following molecule. (CH3)2C═CH(CH2)4CH3arrow_forwardDetermine the number of lone pairs on the nitrogen atom in the following structures.arrow_forwardDraw two constitutional isomers that share the molecular formula C3H,CI. Your structures will have the same molecular formula but will have different connectivities.arrow_forward
- Clearly draw all lone pairs of electrons. onto the following Molecule HOH H₂N мне 0arrow_forwardProvide the correct IUPAC name for the skeletal (line-bond) structure shown here. * 2,2,3,3-3,3- 2,3- 2,2,2,2- 3,3,3,3- tri di penta tetra eth hex meth 2,2-arrow_forwardChoose the condensed structure for the following line structure. CH3C(CH3)2CH₂O(CH2)4CH3 CH3CH₂C(CH3)2CHO(CH₂)4CH3 CH3CH₂C(CH3)2CH₂O(CH₂)4CH3 CH,CH CH(CH,)CH,O(CH,),CH; CH3CH₂C(CH3)2CH₂OCH(CH2)3CH3arrow_forward
- For the following compound identify the lone pairs and indicate if each lone pair is localized or delocalized. Draw out the resonance structures.arrow_forwardDraw all the isomers of C4H10O using bond-line formulas.arrow_forwardIsomers are compounds having different structures but same molecular formula O different rings different molecular formula same structures but different molecular formulaarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





