
Concept explainers
Using the method outlined in Section
alkyl groups, and classify each one as primary, secondary, or tertiary:
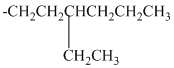
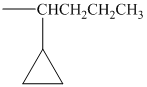
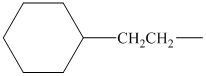
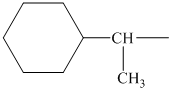
Trending nowThis is a popular solution!

Chapter 2 Solutions
EBK ORGANIC CHEMISTRY
- Give the complete IUPAC name for each of the following compounds: (a) CH3CH2CBr2CH3 (b) (CH3)3CCl (c) (d) CH3CH2CCHCH3CH2CCH (e) (f) (g) (CH3)2CHCH2CH=CH2arrow_forwardGive the complete IUPAC name for each of the following compounds: (a) (CH3)2CHF. (b) CH3CHClCHClCH3 (C) (d) CH3CH2CH = CHCH3 (e) (f) (CH3)3CCH2CCHarrow_forwardDodecane, C12H26, is an unbranched alkane Predict the following: Will it dissolve in water? Will it dissolve in hexane? Will it burn when ignited? Is it a liquid, solid, or gas at room temperature and atmospheric pressure? Is it more or less dense than water?arrow_forward
- Petroleum is a mixture of hydrocarbons made out of decay microscopic ocean-dwelling plants and animals. Crude oil which is the product collects in underground pockets in sedimentary rock can be separated to different fractions by fractional distillation. As a chemical engineer, you are required to prepare the specific details of the products. Two fractions are produced from molecular range C1-C4 and C5 - C12. For each molecular range write the structures for 5 different compounds, formulas and also contain functional group for the IUPAC name of each compound and then identify 2 different functional groups among the structures. Next, provide 5 structures using the dash format, condensed format, bond-line format and full three-dimensional format. Describe the physical properties of the 5 compounds . Determine the rank in increasing order and explain on the basis of intermolecular forces and polarity.arrow_forwardMolecular formula of the given structure is C5H12 Select one: True Falsearrow_forwardUsing Newman projections, draw the most stable conformer for each of the following: a. 3-methylpentane, viewed along the C@2-C@3 bondb. 3-methylhexane, viewed along the C@3-C@4 bondc. 3,3-dimethylhexane, viewed along the C@3-C@4 bondarrow_forward
- A2 1. An alkyne with molecular formula C5H10 2. A ketone with molecular formula C4H8O 3. A ketone with molecular formula C3H8O 4. An alkene with molecular formula C5H8 5. An alkene with molecular formula C5H10 6. An aldehyde with molecular formula C2H4O 7. An aldehyde with molecular formula CH4O 8. A saturated hydrocarbon with molecular formula C6H14arrow_forward2 Q: Consider the following compound: CH3-CH2-CO-CH3 a. suggest a method for increasing the number of carbons by one: b. suggest a method for decreasing the number of carbons by one:arrow_forwardClassify each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°).(a) butane (b) isobutane (c) 2-methylbutane(d) cyclohexane (e) norbornane (bicyclo[2.2.1]heptane)arrow_forward
- How many secondary (2) degree carbons are found in 5 ethyl- 3,3,4 trimethylheptanearrow_forwardThe following molecule contains two functional groups, a haloalkane group and an alkyne. CH3—CHBr—CH=CH2 A) True B) Falsearrow_forwardWhich one of the compounds in Figure 2 is numbered correctly when you need to give the IUPAC name? * 1- A 2- B 3- C 4- Darrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





