
Concept explainers
- a. List the following alcohols in order from strongest acid to weakest acid:
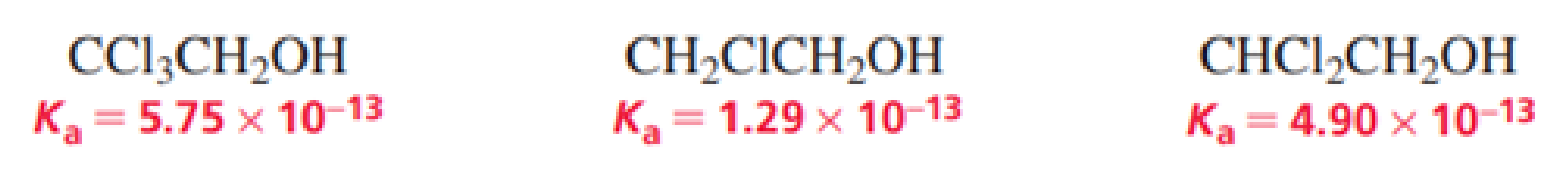
- b. Explain the relative acidities.
(a)
Interpretation:
The given alcohols have to be ranked from strongest to weakest acid.
Concept introduction:
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
If an acid lose one proton, then the formed species is a conjugated base. Weak base forms stronger conjugated acid.
Acidity of species depends on the electronegativity of atom attached to the acidic proton. Order of electronegativity of hybridization is
Acid dissociation constant
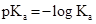
The strength of acid increases as the value of
Answer to Problem 38P
The given alcohols are ranked from strongest to weakest acid as follows,
Explanation of Solution
In hydrocarbons, if hydrogen atoms are replaced by electronegative atoms, it causes inductive electron withdrawal. It stabilizes its conjugate base thus increases the strength of the acid. The conjugated base of a weak acid is very strong. As the electronegativity of substituent increases, the greater will be the inductive electron withdrawal of the substituent making it a strong acid.
Therefore, the acidity order is:
The compound with three chlorine atoms near to the
The alcohol with high
(b)
Interpretation:
The relative acidities of the given alcohol compounds have to be explained briefly.
Concept introduction:
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
If an acid lose one proton, then the formed species is a conjugated base. Weak base forms stronger conjugated acid.
Acidity of species depends on the electronegativity of atom attached to the acidic proton. Order of electronegativity of hybridization is
Explanation of Solution
In hydrocarbons, if hydrogen atoms are replaced by electronegative atoms, it causes inductive electron withdrawal. It stabilizes its conjugate base thus increases the strength of the acid. The electron density near
Want to see more full solutions like this?
Chapter 2 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- Problem 2.32 Problem 2.33 Problem 2.34 For each reaction, label the Lewis acid and base. Use curved arrow notation to show the movement of electron pairs. a. F F 1 B F CH3 CH3 + BBr 3 F _1 b. F-B-O: T F :OH ОН Draw the products of each reaction, and label the nucleophile and electrophile. FO: + AICI 3 CH 3 CH3 Draw the product formed when (CH3CH₂)3N:, a Lewis base, reacts with each Lewis acid: (a) B(CH3)3 (b) (CH3)3C; (c) AICI 3.arrow_forward4. Explain the following.arrow_forwardAnswer the following questions about the four species A–D.a.Which two species represent a conjugate acid–base pair? b. Which two species represent resonance structures? c. Which two species represent constitutional isomers?arrow_forward
- The boiling points of a range of organic compounds (A-D) of similar Mol. Wt.s are given below. OH OMe A в D b.p. 46-50°C Explain why the boiling point of C is higher than that of D. Explain why the boiling point of B is higher than that of A. Explain why the boiling point of C is higher than that of A. Which of these four compounds is most acidic? What makes it so acidic? b.p. 78'C b.p. 141'C b.p. 79'C (i) (ii) (iii) (iv)arrow_forwardGive correct detailed Solutionarrow_forward1. b. Which is a stronger base? 2. orarrow_forward
- Problem 2.5 Problem 2.6 Decide which compound is the acid and which is the base, and draw the products of each proton transfer reaction. a. b. CI a. CI CI OH + OCH 3 -H + H: OH C. b. d. -NH₂ + Draw the products formed from the acid-base reaction of HCI with each compound. HCI OH + H₂SO4 d. NHarrow_forwardAmong the following, which is least acidic? a. p-nitrophenol b. o-cresol С. p-chlorophenol d. phenolarrow_forwardArrange the compounds in order of decreasing basicity A. ethylamine B. 2-aminoethanol C. 3-amino-1-propanolarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
