
Concept explainers
(a)
Interpretation: The orbitals are used to form the indicated bonds in
Concept introduction:
Answer to Problem 20.1P
The orbitals are used to form bond
Explanation of Solution
The given structure of
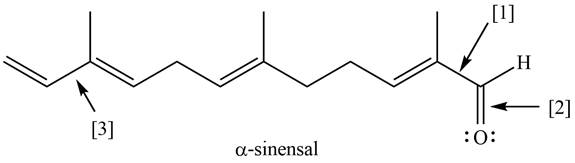
Figure 1
Bond
Bond
Bond
The orbitals are used to form bond
(b)
Interpretation: The type of orbitals where the lone pairs on
Concept introduction:
Answer to Problem 20.1P
The type of orbitals where the lone pairs on
Explanation of Solution
The oxygen atom of carbonyl carbon in
The type of orbitals where the lone pairs on
Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY-STUDY GDE...-W/ACCESS
- ) How many stereogenic double bonds are in octa-1,3,5-triene? How many stereocentersare there?arrow_forwardWhich hydrogens whose bond to carbon is aligned for effective hyperconjugation with the vacant p orbital on the positively charged carbon?arrow_forwardThe double bond of an enamine (alkene + amine) is much more nucleophilic than a typical alkene double bond. Assuming that the nitrogen atom in an enamine is sp2hybridized, draw an orbital picture of an enamine, and explain why the double bond is electron-rich.arrow_forward
- Explain why α-pyrone reacts with Br2 to yield a substitution product (like benzene does), rather than an addition product to one of its C=C bonds.arrow_forwardRank the following groups in order of decreasing priority. −F, −NH2, −CH3, −OHarrow_forwarda. For the following compounds, which pairs of hydrogens (Ha and Hb) are enantiotopic hydrogens?b. Which pairs are diastereotopic hydrogens?arrow_forward
- Is the molecular orbital diagram of 1,3-butadiene the same as cyclopenta-1,3-diene? If so how come? Doesn't the latter have 5 p orbitals or will it technically only have 4?arrow_forwardFor the compounds given (A, B, C) 1. determine their relationship ( enantimers, idential/same compind, diatermeros, constitutional isomers) and give a short explanation whyarrow_forwardFor the following compounds, which pairs of hydrogens (Ha and Hb) are enantiotopic hydrogens? b. Which pairs are diastereotopic hydrogens?arrow_forward
- Explain why alkene A is more stable than alkene B, even though Bcontains more carbon atoms bonded to the double bond. Would youexpect C to be more or less stable than A and B?arrow_forwardHow many rings and π(pi) bonds are contained in compound A and draw one possible structure for this compound A. Compound A has molecular formula C6H10 and is hydrogenated to a compound having molecular formula C6H12arrow_forwardThe trans diaxial geometry for the E2 elimination in chlorocyclohexane. Define this ?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

