
Concept explainers
(a)
Interpretation:
The product for the reaction between
Concept introduction:
The reaction in which one form of an ester is converted to another form by changing the carbon skeleton of the alkoxide part (OR) of the ester is called transesterification. The transesterification reaction is a nucleophilic addition-elimination reaction. In the first step, the nucleophile adds the electro-poor carbonyl carbon of the ester, which forces the bonding pair of carbonyl p bond onto the O atom. A negative charge is produced on that O, and a tetrahedral intermediate is formed. In the second step, tetrahedral intermediate undergoes nucleophilic elimination to form the transesterification product. The transesterification is a reversible reaction. The base hydrolysis of an ester to produce corresponding
Answer to Problem 20.28P
The product for the reaction between
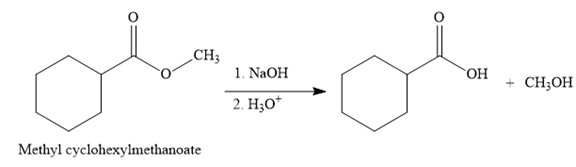
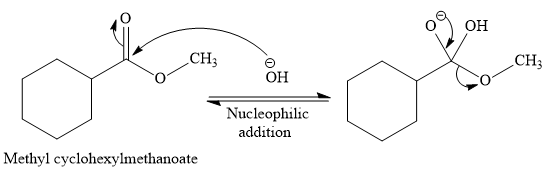
The complete detailed, mechanism for the given reaction is
Explanation of Solution
The given reaction is
So this is a saponification reaction of
In the second step, nucleophilic elimination takes place to produce an alkoxide ion and corresponding carboxylic acid. These steps are reversible.
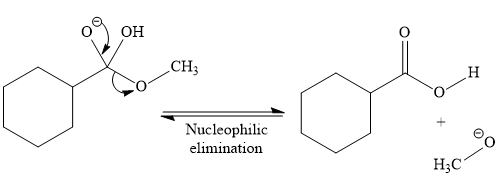
In the third step, irreversible deprotonation of the carboxylic acid takes place and the carboxylate ion is formed. Finally, on acid workup step, protonation of the carboxylate ion takes place to form the carboxylic acid.
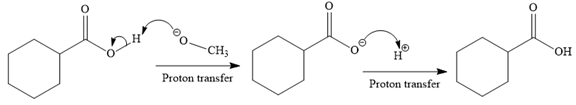
Thus, the products of the given reaction are
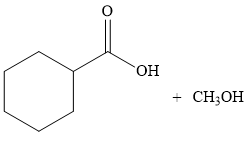
The product is predicted by using transesterification concept and the detailed mechanism is drawn.
(b)
Interpretation:
The product for the reaction between
Concept introduction:
The reaction in which one form of an ester is converted to another form by changing the carbon skeleton of the alkoxide part (OR) of the ester is called transesterification. The transesterification reaction is a nucleophilic addition-elimination reaction. In the first step, the nucleophile adds the electro-poor carbonyl carbon of the ester, which forces the bonding pair of carbonyl p bond onto the O atom. A negative charge is produced on that O, and a tetrahedral intermediate is formed. In the second step, tetrahedral intermediate undergoes nucleophilic elimination to form the transesterification product. The transesterification is a reversible reaction. The base hydrolysis of an ester to produce corresponding carboxylic acid and alcohol is called a saponification reaction.
Answer to Problem 20.28P
The product for the reaction between
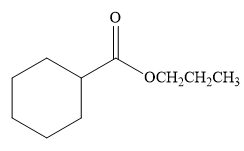
The complete detailed, mechanism for the given reaction is
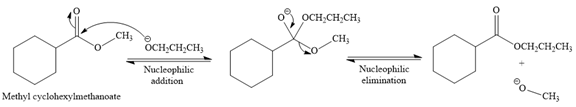
Explanation of Solution
The given reaction is
This is a transesterification reaction of
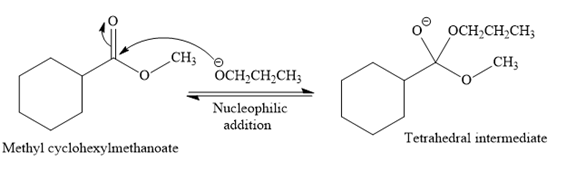
In the second step, the tetrahedral intermediate undergoes nucleophilic elimination to form the transesterification product.
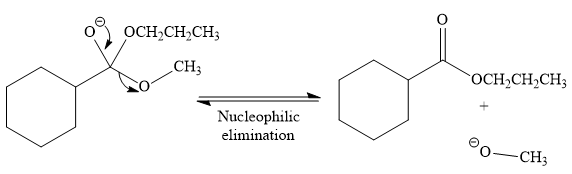
Thus, the product of the given reaction is

The product is predicted by using transesterification concept and the detailed mechanism is drawn.
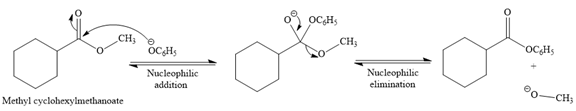
(c)
Interpretation:
The product for the reaction between
Concept introduction:
The reaction in which one form of an ester is converted to another form by changing the carbon skeleton of the alkoxide part (OR) of the ester is called transesterification. The transesterification reaction is a nucleophilic addition-elimination reaction. In the first step, the nucleophile adds the electro-poor carbonyl carbon of the ester, which forces the bonding pair of carbonyl p bond onto the O atom. A negative charge is produced on that O, and a tetrahedral intermediate is formed. In the second step, tetrahedral intermediate undergoes nucleophilic elimination to form the transesterification product. The transesterification is a reversible reaction. The base hydrolysis of an ester to produce corresponding carboxylic acid and alcohol is called a saponification reaction.
Answer to Problem 20.28P
The product for the reaction between
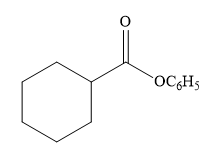
The complete detailed, mechanism for the given reaction is
Explanation of Solution
The given reaction is
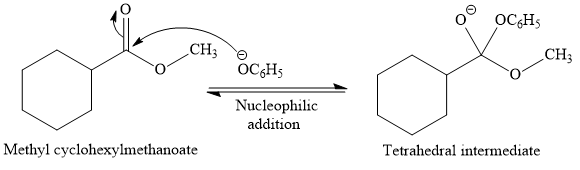
This is a transesterification reaction of the
In the second step, tetrahedral intermediate undergoes nucleophilic elimination to form the transesterification product.
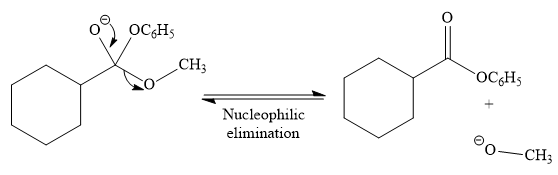
Since phenoxide ion (
Thus, the product of the given reaction is

The product is predicted by using transesterification concept and the detailed mechanism is drawn.
Want to see more full solutions like this?
Chapter 20 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- (a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C=C and an OH group. 2-methylcyclohexanonearrow_forwardSynthesize the products by drawing out reagents and intermediates along the way.arrow_forwardParts A,B&C Please.arrow_forward
- Provide structure(s) for the starting material(s), reagent(s) or the major organic product(s) of each of the following reactions.arrow_forwardDraw the structure of each of the following molecules. (a) cyclohexyl butanoate; (b) 1,1-dimethylethyl hexanoate; (c) phenyl 4,4-dinitroheptanoatearrow_forwardAn important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and its alkyl-substituted derivative: Base CH;CH2O OCH,CH3 CH;CH,0 OCH2CH3 H2 Diethyl malonate Base CH;CH,0 °C `OCH,CH3 CH;CH,O OCH,CH3 R Alkyl substituted diethyl malonate NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is typically used to deprotonate the alkyl-substituted derivative. Explain why.arrow_forward
- Question:In organic chemistry, the formation of carbocations is a critical step in many reactions. Consider the following scenario: A tertiary alcohol, 2-methyl-2-butanol, undergoes acid-catalyzed dehydration to form an alkene. However, when this reaction is attempted using concentrated sulfuric acid, an unexpected product is obtained. Explain the factors responsible for this unexpected product formation and propose a mechanism for the reaction.arrow_forwardDraw the structure of the following compounds which parent names have been traced to a common name; (a)5-methyl-4-nitroimidazole (b)2-chloro-4-methoxythiazole.arrow_forwardDetermine the major product that is formed when the alkyl halide reacts with a hydroxide ion in an elimination reaction. b) For the major elimination product obtained in 5a), which stereoisomer (cis or trans) is obtained in greater yield? Draw the two isomers and provide the names of the compounds.arrow_forward
- Draw the alcohol you’d oxidize to produce (a) 2-methyl-propanal; (b) 2-pentanone; (c) 3-methylbutanoic acid.arrow_forwardcomplete the synthesis cyclohexane to 1R,2R 2 bromocyclohexanolarrow_forward(b) Consider the reaction of 1-bromobutane with a large excess of ammonia (NH3). Draw the reactants, the transition state, andthe products. Note that the initial product is the salt of an amine (RNH3+ Br - ), which is deprotonated by the excess ammonia to give the amine.arrow_forward
