
Concept explainers
(a)
Interpretation:
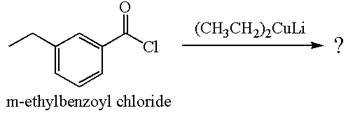
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to corresponding ketone by reacting it with lithium dialkylcuprate
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
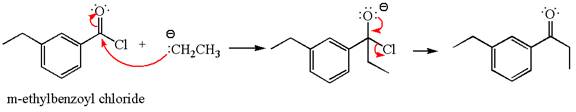
Explanation of Solution
The equation for the reaction of
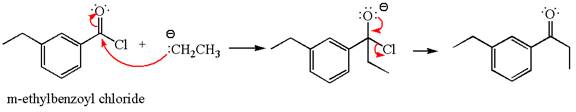
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(b)
Interpretation:
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to an
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
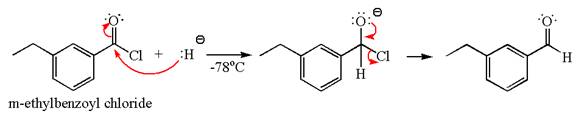
Explanation of Solution
The equation for the reaction of
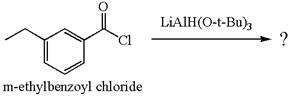

The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(c)
Interpretation:
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to a primary alcohol by reacting with
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
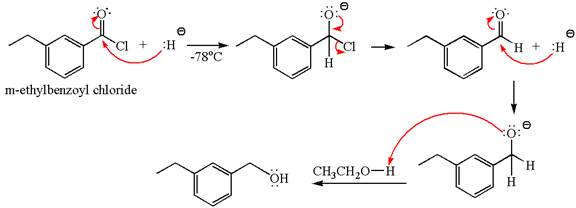
Explanation of Solution
The equation for the reaction of
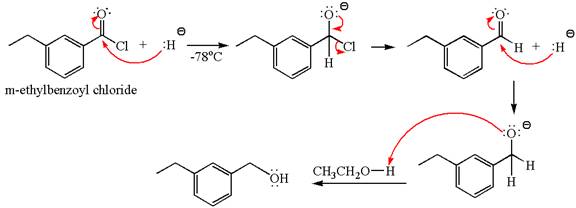
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(d)
Interpretation:
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to a tertiary alcohol by reacting it with
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
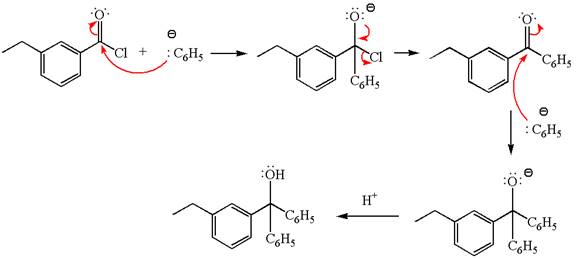
Explanation of Solution
The equation for the reaction of
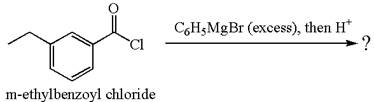

The product with detailed mechanism for the given reaction is drawn based on the reactivity of
Want to see more full solutions like this?
Chapter 20 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 22). What is the major product of the following reactions? (A) (B) Br 1) NaCN 2) CH3MgBr 3) H30*/H₂O (C) LOH (D) CNarrow_forwardDraw the products formed when benzoyl chloride (C,H;COCI) is treated with each nucleophile: (a) H20, pyridine; (b) CH;CO0"; (c) NH3 (excess); (d) (CHaNH (excess).arrow_forward1,2-Diols are converted to carbonyl compounds when treated with strong acids, in a reaction called the pinacol rearrangement. (a) Draw a stepwise mechanism for this reaction. (Hint: The reaction proceeds by way of carbocation intermediates.) (b) Assuming that the pinacol rearrangement occurs via the more stable carbocation, draw the rearrangement product formed from diol D.arrow_forward
- Which of the following compounds will form a yellow solid when dissolved in a basic, aqueous solution of I2?(a) butanoic acid; (b) pentan-2-one; (c) pentan-3-one; (d) cyclohexanone; (e) pentanalarrow_forwarda) Provide a mechanism that accounts for the product formed in the following reaction. OH CH H" ► 1-ethoxy-1-methyleyclohexane + CH,CH,OH b) Show the structure of the product(s) formed in the following reaction and write the mechanism for the reaction. OH CH + (CH;);COHarrow_forward1,2-Diols are converted to carbonyl compounds when treated with strong acids, in a reaction called the pinacol rearrangement. (a) Draw a stepwise mechanism for this reaction. (Hint: The reaction proceeds by way of carbocation intermediates.) (b) Assuming that the pinacol rearrangement occurs via the more stable carbocation, draw the rearrangement product formed from diol D.arrow_forward
- (a) Draw the mechanism for the formation of both of the enols that can be formed from A (use acetic acid & AcOH as the source of the protons) (b) Draw the mechanism of reaction of this enol with bromine to give product Barrow_forwardWhat reaction took place in the reaction below? CHẠCH, CH; CH,(CH),CH; CH{CH,),C-OCH; CHẠCH)A CH; CHỊCHCo H0 НОСН CH/CH),CH, CH, (CH),C-C HOCH, HH O reduction (hydrogenation) O amide synthesis O acetal hydrolysis O ester synthesis O acid base O dehydration O thioester synthesis O phosphate ester synthesis O hydration O ester hydrolysis O amide hydrolysis O oxidation O acetal synthesisarrow_forwardDevise at least three different methods to prepare N-methylbenzylamine (PhCH2NHCH3) from benzene, any one-carbon organic compounds, and any required reagents.arrow_forward
- Draw the structure of each of the following molecules. (a) cyclohexyl butanoate; (b) 1,1-dimethylethyl hexanoate; (c) phenyl 4,4-dinitroheptanoatearrow_forwardQuestion attachedarrow_forwardDraw the structure of each of the following molecules (a) 2,2-dimethylcyclopentane-1-carboxylic acid;(b) (R)-3-chloropentanoic acid; (c) (2R,3S)-2,3-dinitrobutanedioic acidarrow_forward
