
Concept explainers
(a)
Interpretation: The product formed when the
Concept introduction: The substitution reaction involves the replacement of one functional group by other functional group. In nucleophilic substitution an electron rich species attack the species that is deficient in electrons. The electrophile and the leaving group together form a substrate. The nucleophile attacks over the substrate and there occurs the removal of leaving group from the substrate.
Answer to Problem 20.41P
The product formed when the
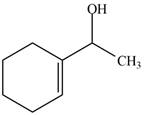
Figure 1
Explanation of Solution
The reaction that shows the formation of product when
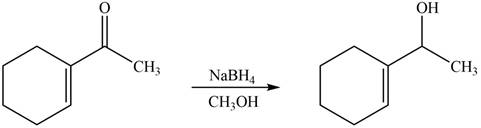
Figure 2
In the given reaction, sodium borohydride is used as a reducing agent. Sodium borohydride is used for the reduction of carbonyl compounds to alcohols.
The product formed when the
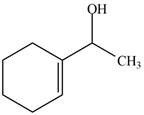
Figure 1
(b)
Interpretation: The product formed when the
Concept introduction: Addition of hydrogen takes place across a double bond of
Answer to Problem 20.41P
The product formed when the
Figure 3
Explanation of Solution
The reaction that shows the formation of product when
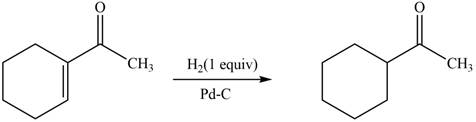
Figure 4
In the given reaction, hydrogenation of alkene takes place in the presence of palladium.
The product formed when the
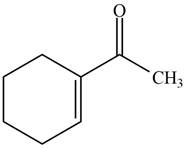
Figure 3
(c)
Interpretation: The product formed when the
Concept introduction: Addition of hydrogen takes place across a double bond of alkenes to form alkanes. This process is known as hydrogenation. It takes place in the presence of catalysts such as nickel, platinum or palladium.
Answer to Problem 20.41P
The product formed when the
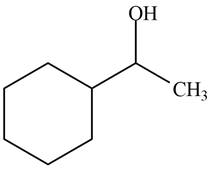
Figure 5
Explanation of Solution
The reaction that shows the formation of product when
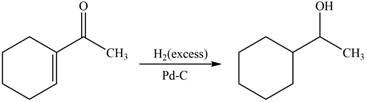
Figure 6
In the given reaction, excess hydrogen is used which will reduce both keto group and double bond. This results in the formation of a saturated compound.
The product formed when the

Figure 5
(d)
Interpretation: The product formed when the
Concept introduction: Methyllithium in the presence of water reduces carbonyl compounds into alcohol. Methyllithium is a reducing agent.
Answer to Problem 20.41P
The product formed when the
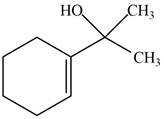
Figure 7
Explanation of Solution
The reaction that shows the formation of product when
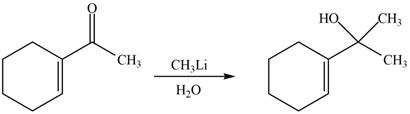
Figure 8
The product formed in the given reaction is
The product formed when the
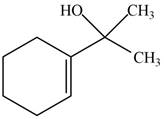
Figure 7
(e)
Interpretation: The product formed when the
Concept introduction: Grignard reagent is prepared by the reaction of alkyl or aryl bromide with magnesium metal in the presence of ether. The reaction of Grignard reagent with an
Answer to Problem 20.41P
The product formed when the
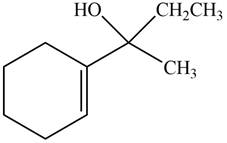
Figure 9
Explanation of Solution
The reaction that shows the formation of product when
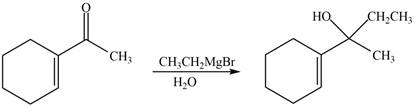
Figure 10
In the given reaction, Grignard reagent is used. This reagent attacks on the carbonyl compound to form alkoxides. In the next step, protonation with water takes place to give the final product.
The product formed when the
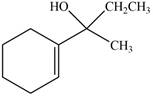
Figure 9
(f)
Interpretation: The product formed when the
Concept introduction: The steps involved in the reaction of ketone with organolithium reagent are as follows:
• The first step involves the reduction of carbonyl group into single bonds.
• The second step is the addition of R group from organometallic reagent and hydrogen to oxygen.
Answer to Problem 20.41P
The product formed when the
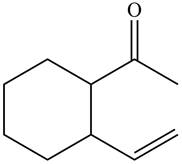
Figure 11
Explanation of Solution
The reaction that shows the formation of product when
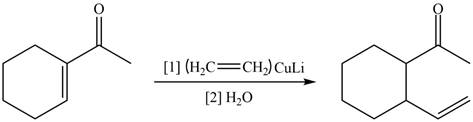
Figure 12
In the given reaction, organo copper reagent reacts with unsaturated carbonyl compound and forms carbon–carbon double bond with the beta carbon as shown in the above reaction.
The product formed when the

Figure 11
Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY W/ ACCESS >IC<
- Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs. [1] C6H5Li (excess); [2] H2Oarrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] HC≡CNa; [2] H2Oarrow_forwardDraw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs.a. NaBH4, CH3OHb. [1] LiAlH4; [2] H2Oc. [1] CH3MgBr (excess); [2] H2Od. [1] C6H5Li (excess); [2] H2Oe. Na2Cr2O7, H2SO4, H2Oarrow_forward
- Draw the product formed when (CH3)2CHOH is treated with each reagent. a.SOCl2, pyridine b. TsCl, pyridine c.H2SO4 d.HBr e.PBr3, then NaCN f.POCl3, pyridinearrow_forwardDraw the product formed when (CH3)2CHOH is treated with each reagent. a. SOCl2, pyridine b. TsCl, pyridine c. H2SO4 d. HBr e. PBr3, then NaCN f. POCl3, pyridinearrow_forward1. Draw the product formed when phenylacetaldehyde (C6H5CH2CHO) is treated with each reagent:arrow_forward
- Draw the product formed when (CH3)2CHOH is treated with each reagent (a, b and c)arrow_forwardDraw the products when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents no reaction occurs: A)NaCl B)NH3(1 eqiv) C)1)CH2NH2, 2)CH3COClarrow_forwardDraw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then H2O;(b) NaBH4 in CH3OH;(c) H2 (1 equiv), Pd-C;(d) H2 (excess), Pd-C;(e) NaBH4 (excess) in CH3OH;(f) NaBD4 in CH3OH.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





