
Fundamentals of Physics, Volume 1, Chapter 1-20
10th Edition
ISBN: 9781118233764
Author: David Halliday
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 20, Problem 35P
The cycle in Fig. 20-31 represents the operation of a gasoline internal combustion engine. Volume V3 = 4.00V1. Assume the gasoline-air intake mixture is an ideal gas with γ = 1.30. What are the ratios (a) T2/T1,(b) T3/T1, (c) T4/T1, (d) p3/p1, and (e) p4/p1? (f) What is the engine efficiency?
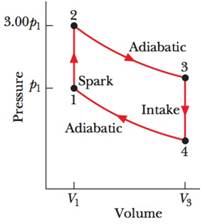
Figure 20-31 Problem 35.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Two containers each hold 1 mole of an ideal gas at 1 atm. Container A holds a monatomic gas and container B holds a diatomic gas. The volume of each container is halved while the pressure is held constant. (Assume the initial volumes of containers A and B are equal.)
(c)
What is the ratio
QA
QB
of the energy transferred to gases A and B?
It's a thermodynamics question.
A cylinder of volume 0.290 m3 contains 12.0 mol of neon gas at 19.2°C. Assume neon behaves as an ideal gas.
(a) What is the pressure of the gas? Pa(b) Find the internal energy of the gas. J(c) Suppose the gas expands at constant pressure to a volume of 1.000 m3. How much work is done on the gas? J(d) What is the temperature of the gas at the new volume? Your response differs from the correct answer by more than 10%. Double check your calculations. K(e) Find the internal energy of the gas when its volume is 1.000 m3. J(f) Compute the change in the internal energy during the expansion. J(g) Compute ΔU − W. J(h) Must thermal energy be transferred to the gas during the constant pressure expansion or be taken away?
This answer has not been graded yet.
(i) Compute Q, the thermal energy transfer. J(j) What symbolic relationship between Q, ΔU, and W is suggested by the values obtained?
Chapter 20 Solutions
Fundamentals of Physics, Volume 1, Chapter 1-20
Ch. 20 - Point i in Fig. 20-19 represents the initial state...Ch. 20 - In lour experiments, blocks A and B, starting ill...Ch. 20 - A gas, confined to an insulated cylinder, is...Ch. 20 - An ideal monatomic gas at initial temperature T0...Ch. 20 - In four experiments, 2.5 mol of hydrogen gas...Ch. 20 - A box contains 100 atoms in a configuration that...Ch. 20 - Does the entropy per cycle increase, decrease, or...Ch. 20 - Three Carnot engines operate between temperature...Ch. 20 - An inventor claims to have invented four engines,...Ch. 20 - Does the entropy per cycle increase, decrease, or...
Ch. 20 - SSM Suppose 4.00 mol of an ideal gas undergoes a...Ch. 20 - An ideal gas undergoes a reversible isothermal...Ch. 20 - ILW A 2.50 mol sample of an ideal gas expands...Ch. 20 - How much energy must be transferred as heat for a...Ch. 20 - ILW Find a the energy absorbed as heat and b the...Ch. 20 - a What is the entropy change of a 12.0 g ice cube...Ch. 20 - ILW A 50.0 g block of copper whose temperature is...Ch. 20 - At very low temperatures, the molar specific heat...Ch. 20 - A 10 g ice cube at 10oC is placed in a lake whose...Ch. 20 - A 364 g block is put in contact with a thermal...Ch. 20 - SSM WWW In an experiment, 200 g of aluminum with a...Ch. 20 - A gas sample undergoes a reversible isothermal...Ch. 20 - In the irreversible process of Fig. 20-5, let the...Ch. 20 - Prob. 14PCh. 20 - A mixture of 1773 g of water and 227 g of ice is...Ch. 20 - GO An 8.0 g ice cube at -10C is put into a Thermos...Ch. 20 - Prob. 17PCh. 20 - GO A 2.0 mol sample of an ideal monatomic gas...Ch. 20 - Suppose 1.00 mol of a monatomic ideal gas is taken...Ch. 20 - Expand 1.00 mol of an monatomic gas initially at...Ch. 20 - GO Energy can be removed from water as heat at and...Ch. 20 - GO An insulated Thermos contains 130 g of water at...Ch. 20 - A Carnot engine whose low-temperature reservoir is...Ch. 20 - A Carnot engine absorbs 52 kJ as heat and exhausts...Ch. 20 - A Carnot engine has an efficiency of 22.0. It...Ch. 20 - In a hypothetical nuclear fusion reactor, the fuel...Ch. 20 - SSM WWW A Carnot engine operates between 235C and...Ch. 20 - In the first stage of a two-stage Carnot engine,...Ch. 20 - GO Figure 20-27 shows a reversible cycle through...Ch. 20 - A 500 W Carnot engine operates between...Ch. 20 - The efficiency of a particular car engine is 25...Ch. 20 - GO A Carnot engine is set up to produce a certain...Ch. 20 - SSM ILW Figure 20-29 shows a reversible cycle...Ch. 20 - GO An ideal gas 1.0 mol is the working substance...Ch. 20 - The cycle in Fig. 20-31 represents the operation...Ch. 20 - How much work must be done by a Carnot...Ch. 20 - SSM A heat pump is used to heal a building, The...Ch. 20 - The electric motor of a heat pump transfers energy...Ch. 20 - SSM A Carnot air conditioner lakes energy from the...Ch. 20 - To make ice, a freezer that is a reverse Carnot...Ch. 20 - ILW An air conditioner operating between 93F and...Ch. 20 - The motor in a refrigerator has a power of 200 W....Ch. 20 - GO Figure 20-32 represents a Carnot engine that...Ch. 20 - a During each cycle, a Carnot engine absorbs 750 J...Ch. 20 - Prob. 45PCh. 20 - A box contains N identical gas molecules equally...Ch. 20 - SSM WWW A box contains N gas molecules, Consider...Ch. 20 - Four particles are in the insulated box of Fig....Ch. 20 - A cylindrical copper rod of length 1.50 m and...Ch. 20 - Suppose 0.550 mol of an ideal gas is isothermally...Ch. 20 - Prob. 51PCh. 20 - Suppose 1.0 mol of a monatomic ideal gas initially...Ch. 20 - GO Suppose that a deep shaft were drilled in...Ch. 20 - What is the entropy change for 3.20 mol of an...Ch. 20 - A 600 g lump of copper at 80.0C is placed in 70.0...Ch. 20 - Figure 20-33 gives the force magnitude F versus...Ch. 20 - The temperature of 1.00 mol of a monatomic ideal...Ch. 20 - Repeat Problem 57, with the pressure now kept...Ch. 20 - SSM A 0.600 kg sample of water is initially ice at...Ch. 20 - A three-step cycle is undergone by 3.4 mol of an...Ch. 20 - An inventor has built an engine X and claims that...Ch. 20 - Suppose 2.00 mol of a diatomic gas is taken...Ch. 20 - A three-step cycle is undergone reversibly by 4.00...Ch. 20 - a A Carnot engine operates between a hot reservoir...Ch. 20 - A 2.00 mol diatomic gas initially at 300 K...Ch. 20 - An ideal refrigerator does 150 J of work to remove...Ch. 20 - Suppose that 260 J is conducted from a...Ch. 20 - An apparatus that liquefies helium is in a room...Ch. 20 - GO A brass rod is in thermal contact with a...Ch. 20 - A 45.0 g block of tungsten at 30.0C and a 25.0 g...Ch. 20 - Prob. 71PCh. 20 - Calculate the efficiency of a fossil-fuel power...Ch. 20 - SSM A Carnot refrigerator extracts 35.0 kJ as heat...Ch. 20 - A Carnot engine whose high-temperature reservoir...Ch. 20 - SSM System A of three particles and system B of...Ch. 20 - Figure 20-36 shows a Carnot cycle on a T-S...Ch. 20 - Find the relation between the efficiency of a...Ch. 20 - A Carnot engine has a power of 500 W. It operates...Ch. 20 - In a real refrigerator, the low-temperature coils...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The required force by second crewman so that the net force of the two crewmen is in the forward direction.
Physics (5th Edition)
The sum of the series ∑n=1∞2nn! , use the Maclaurin series.
Mathematical Methods in the Physical Sciences
In which situation shown below will the electromagnetic wave be more successful in inducing a current in the lo...
University Physics Volume 2
52. You are target shooting using a toy gun that fires a small ball at a speed of 15 m/s. When the gun is fire...
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
A dipole of moment p = 2.9 nC m consists of two charges separated by far less than 10 cm. Find the potential 1...
Essential University Physics: Volume 2 (3rd Edition)
72. Magma is generated at divergent and convergent plate boundaries. What type of magma is dominant at each bou...
Conceptual Physical Science (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A cylinder of volume 0.320 m3 contains 10.5 mol of neon gas at 17.4°C. Assume neon behaves as an ideal gas. (a) What is the pressure of the gas? Pa(b) Find the internal energy of the gas. J(c) Suppose the gas expands at constant pressure to a volume of 1.000 m3. How much work is done on the gas? J(d) What is the temperature of the gas at the new volume? K(e) Find the internal energy of the gas when its volume is 1.000 m3. J(f) Compute the change in the internal energy during the expansion. J(g) Compute ΔU − W. J(h) Must thermal energy be transferred to the gas during the constant pressure expansion or be taken away? This answer has not been graded yet. (i) Compute Q, the thermal energy transfer. J(j) What symbolic relationship between Q, ΔU, and W is suggested by the values obtained?arrow_forwardA cylinder containing a system of gas is taken through the cycle as shown in Figure 2, calculate a) the work done by the gas during this process ABCD, b) the work input (work done on the gas) during the process DA, e the change in the internal energy of the gas during the cycle, and d) the net energy input Q during this cycle. P((kPa) 50 40 30 20 A 10 3. V (m) Figure 1: Thermodynamic process for Question 5arrow_forwardA cylinder of volume 0.260 m3 contains 11.2 mol of neon gas at 22.4°C. Assume neon behaves as an ideal gas. (a) What is the pressure of the gas? Pa(b) Find the internal energy of the gas. J(c) Suppose the gas expands at constant pressure to a volume of 1.000 m3. How much work is done on the gas?J(d) What is the temperature of the gas at the new volume?arrow_forward
- A cylinder of volume 0.300 m3 contains 10.0 mol of neon gas at 20.0°C. Assume neon behaves as an ideal gas. (a) What is the pressure of the gas? (b) Find the internal energy of the gas. (c) Suppose the gas expands at constant pressure to a volume of 1.000 m3. How much work is done on the gas? (d) What is the temperature of the gas at the new volume? (e) Find the internal energy of the gas when its volume is 1.000 m3 . (f) Compute the change in the internal energy during the expansion. (g) Compute ΔU − W (h) Must thermal energy be transferred to the gas during the constant pressure expansion or be taken away? (i) Compute Q, the thermal energy transfer. (j) What symbolic relationship between Q , Δ U , and W is suggested by the values obtained?arrow_forwardA cylinder of volume 0.300 m3 contains 10.0 mol of neon gas at 20.0°C. Assume neon behaves as an ideal gas. (a) What is the pressure of the gas? (b) Find the internal energy of the gas. (c) Suppose the gas expands at constant pressure to a volume of 1.000 m3. How much work is done on the gas? (d) What is the temperature of the gas at the new volume? (e) Find the internal energy of the gas when its volume is 1.000 m3. (f) Compute the change in the internal energy during the expansion. (g) Compute ΔU - W. (h) Must thermal energy be transferred to the gas during the constant pressure expansion or be taken away? (i) Compute Q, the thermal energy transfer. (j) What symbolic relationship between Q, ΔU, and W is suggested by the values obtained?arrow_forwardA sample of ideal gas expands from an initial pressure and volume of 32 atm and 1.0 L to a final volume of 4.0 L. The initial temperature is 300 K. If the gas is monatomic and the expansion isothermal, what are the (a) final pressure pf , (b) final temperature Tf , and (c) work W done by the gas? If the gas is monatomic and the expansion adiabatic, what are (d) pf , (e) Tf , and (f) W? If the gas is diatomic and the expansion adiabatic, what are (g) pf , (h) Tf , and (i) W?arrow_forward
- Question 1. An ideal diatomic gas contracts from 1.25 m³ to 0.500 m³ at a constant pressure of 1.50 x 10°P.. Draw a PV diagram and name this process that occurs at constant pressure. If the initial temperature is 425 K, calculate (a) the work done on the gas, (b) the change in internal energy of the gas, (c) the energy transfer, Q, and, (d) the final temperature.arrow_forwardThe volume of an ideal gas is adiabatically reduced from 200 L to 74.3 L. The initial pressure and temperature are 1.00 atm and 300 K.The final pressure is 4.00 atm. (a) Is the gas monatomic, diatomic, or polyatomic? (b) What is the final temperature? (c) How many moles are in the gas?arrow_forwardProblem 10: In an adiabatic process oxygen gas in a container is compressed along a path that can be described by the following pressure p, in atm, as a function of volume V, in liters:p = p0 V-6/5.Here p0 is a constant of units atm⋅L6/5. Part (a) Write an expression for the work W done on the gas when the gas is compressed from a volume Vi to a volume Vf. Part (b) The initial and final volumes during the process were Vi = 5 L and Vf = (Vi/2) L, respectively. If p0 = 3.5 atm⋅L6/5, find the amount of work done on the gas, in joules.arrow_forward
- Given 1 mol of a monatomic ideal gas and 1 mol of a diatomic ideal gas, it is known that after a process, both gases undergo the same change in internal energy. Regarding the temperature variation, we can say that: a) Nothing can be said because the answer depends on the chemical elements that make up the molecules.b) The temperature change is greater in diatomic gas.c) The temperature variation is greater in monatomic gas.d) The gases undergo the same change in temperature.arrow_forwardProblem 6: A certain gasoline engine is modeled as a monatomic ideal gas undergoing an Otto cycle, represented by the p-V diagram shown in the figure. The initial pressure, volume, and temperature are p1 = 0.95 × 105 Pa, V1 = 0.025 m3, and T1 = 310 K, respectively. Part (a) Calculate the number of moles times the gas constant, nR, in joules per kelvin, to three significant figures, using the ideal-gas law and the initial values of pressure, volume, and temperature. This quantity will be useful for later calculations. Part (b) Calculate the work performed on the gas during the first step, in joules, for V2 = V1/6.2. Part (c) Calculate the temperature of the gas, in kelvins, at the end of the first step. Part (d) The second step in the Otto cycle is isochoric (constant-volume) heating. Calculate the heat absorbed by the gas during this process, in joules, if the temperature is increased so that T3 = 1.53T2. Part (e) Calculate the pressure at the end of the isochoric heating step,…arrow_forwardA cylinder with a piston holds 3.00 moles of a diatomic gas. The gas in the cylinder absorbs 875 J of energy due to the higher temperature of the environment. At the same time, the cylinder expands to a larger volume, doing 137 J of work on the environment. (a) What is the change in internal energy of the gas in the cylinder (in J)? (b) What is the change in temperature of the gas (in K)?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning
The Second Law of Thermodynamics: Heat Flow, Entropy, and Microstates; Author: Professor Dave Explains;https://www.youtube.com/watch?v=MrwW4w2nAMc;License: Standard YouTube License, CC-BY