
(III) The operation of a diesel engine can be idealized by the cycle shown in Fig. 20-18. Air is drawn into the cylinder during the intake stroke (not part of the idealized cycle). The air is compressed adiabatically, path ab. At point b diesel fuel is injected into the cylinder which immediately burns since the temperature is very high. Combustion is slow, and during the first part of the power stroke, the gas expands at (nearly) constant pressure, path bc. After burning, the rest of the power stroke is adiabatic, path ed. Path da corresponds to the exhaust stroke. (a) Show that, for a quasistatic reversible engine undergoing this cycle using an ideal gas, the ideal efficiency is
where
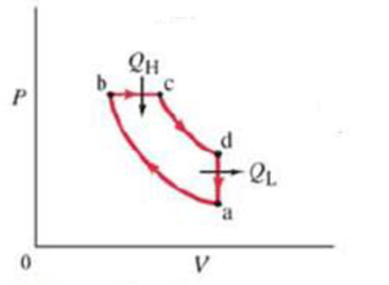
FIGURE 20-18
Problem 7.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
PHYSICS F/SCI.+ENGINEERS-ACCESS
Additional Science Textbook Solutions
Conceptual Integrated Science
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
College Physics (10th Edition)
University Physics (14th Edition)
Cosmic Perspective Fundamentals
College Physics: A Strategic Approach (4th Edition)
- Does the entropy increase for a Carnot engine for each cycle?arrow_forwardThe energy output of a heat pump is greater than the energy used to operate the pump. Why doesn't this statement violate the first law of thermodynamics?arrow_forwardIn an isochoric process, heat is added to 10 mol of monoatomic ideal gas whose temperature increases from 273 to 373 K. What is the entropy change of the gas?arrow_forward
- Is it possible for a system to have an entropy change if it neither absorbs nor emits heat during a reversible? transition? What happens it the process is irreversible?arrow_forwardFor the Carnot cycle of Figure 4.12, what is the entropy change of the hot reservoir, the cold reservoir, and the universe? Figure 4.11 The four processes of the Carnot cycle. The working substance is assumed to be an ideal gas whose thermodynamic path MNOP is represented in Figure 4.12. Figure 4.12 The total work done by the gas in the Carnot cycle is shown and given by the area enclosed by the loop MNOPM.arrow_forwardA Carnot engine operates between 550 and 20 baths and produces 300 kJ of energy in each cycle. Find the change in entropy of the (a) hot bath and (b) cold bath, in each Carnot cycle?arrow_forward
- A 0.50-kg piece of aluminum at 250 is dropped into 1.0 kg of water at 20 . After equilibrium is reached, what is the net entropy change of the system?arrow_forwardSuppose 20 g of ice at 0 is added to 300 g of water at 60 . What is the total change in entropy of the mixture after it reaches thermal equilibrium?arrow_forwardA Carnot cycle working between 100 and 30 is used to drive a refrigerator between 10 and 30 . How much energy must the Carnot engine produce per second so that the refrigerator is able to discard 10 J of energy per second?arrow_forward
- A Carnot engine is used to measure the temperature of a heat reservoir. The engine operates between the heat reservoir and a reservoir consisting of water at its triple point. (a) If 400 J per cycle are removed from the heat reservoir while 200 J per cycle are deposited in the triple-point reservoir, what is the temperature of the heat reservoir? (b) If 400 J per cycle are removed from the triple-point reservoir while 200 J per cycle are deposited in the heat reservoir, what is the temperature of the heat reservoir?arrow_forwardCheck Your Understanding In Example 4.7, the spontaneous flow of heat from a hot object to a cold object results in a net increase in entropy of the universe. Discuss how this result can be related to an increase in disorder of the system.arrow_forwardWhat is the decrease in entropy of 25.0 g of water that condenses on a bathroom mirror at a temperature of 35.0C, assuming no change in temperature and given the latent heat of vaporization to be 2450 kJ/kg?arrow_forward

 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

