
Concept explainers
(a)
Interpretation:
The enantiomer of the given monosaccharide has to be drawn and D or L-sugar has to be identified from the pair.
Concept introduction:
The pair of Enantiomers is non-superimposable mirror images of each other.
The stereo-descriptor used for carbohydrates is D or L. it is based on the dextrorotatory or levorotatory of smallest carbohydrate glyceraldehyde (1 chiral center).
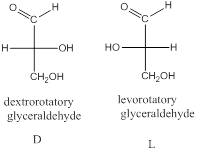
The stereo-descriptor for other carbohydrates of having more than one chiral center will be evaluated by the location of –OH group (right or left) of farthest chiral carbon (bottom) from the carbonyl group. Such as,
If the –OH group is located in right side then, the carbohydrate is a D-sugar.
If the –OH group is located in left side then, the carbohydrate is an L-sugar.
(b)
Interpretation:
The enantiomer of the given monosaccharide has to be drawn and D or L-sugar has to be identified from the pair.
Concept introduction:
The pair of Enantiomers is non-superimposable mirror images of each other.
The stereo-descriptor used for carbohydrates is D or L. it is based on the dextrorotatory or levorotatory of smallest carbohydrate glyceraldehyde (1 chiral center).

The stereo-descriptor for other carbohydrates of having more than one chiral center will be evaluated by the location of –OH group (right or left) of farthest chiral carbon (bottom) from the carbonyl group. Such as,
If the –OH group is located in right side then, the carbohydrate is a D-sugar.
If the –OH group is located in left side then, the carbohydrate is an L-sugar.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Modified Masteringchemistry With Pearson Etext -- Valuepack Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry
- draw the following amino acid chains and give the single and three letter abbreviations for cysteinearrow_forwardDraw the Fischer projection for a monosaccharide.arrow_forwardConstruct the two enantiomeric forms/structure of the following monosaccharides and designate the handedness of each using D, L system: a. Ribulosearrow_forward
- Complete the following table by providing the Fischer and Haworth Projections of the given sugars. In the Fischer Projection put an asterisk (*) mark in the chiral centers of the molecule. Sugar Fischer Projection Haworth Projection (Complete) alpha-anomer Haworth Projection (Abbreviated) beta-anomer L-Galactose L-Fructose D- Xylosearrow_forwardIdentify the monosaccharide represented by each of the following structures.arrow_forwardDraw a ceramide with a (16:1n-7) fatty acid attachedarrow_forward
- Complete the following table by providing the Fischer and Haworth Projections of the given sugars. In the Fischer Projection put an asterisk (*) mark in the chiral centers of the molecule. Sugar Fischer Projection Haworth Projection (Complete) alpha-anomer Haworth Projection (Abbreviated) beta-anomer L-Galactose L-Fructose D-Mannose D-Xylose L-Lyxosearrow_forwardComplete the following table by providing the Fischer and Haworth Projections of the given sugars. In the Fischer Projection put an asterisk (*) mark in the chiral centers of the molecule. Sugar Fischer Projection Haworth Projection (Complete) alpha-anomer Haworth Projection (Abbreviated) beta-anomer D-Mannose D-Xylose L-Lyxosearrow_forwardComplete the following table by providing the Fischer and Haworth Projections of the given sugars. (Please be careful with the alpha and beta, and the complete and abbreviated) (not a graded question) Sugar Fischer Projection Haworth Projection (Complete) alpha-anomer Haworth Projection (Abbreviated) beta-anomer L-Galactose L-Fructosearrow_forward
