
Concept explainers
(a)
Interpretation:
Anomer of the aldohexose should be converted to its Haworth projection.
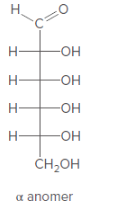
Concept Introduction:
Haworth formula indicates the connectivity of atoms in a ring structure. The linear structure should be used in the conversion.
(b)
Interpretation:
Anomer of the aldohexose should be converted to its Haworth projection.
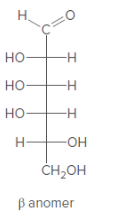
Concept Introduction:
Haworth formula indicates the connectivity of atoms in a ring structure. The linear structure should be used in the conversion.
(c)
Interpretation:
Anomer of the aldohexose should be converted to its Haworth projection.
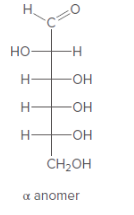
Concept Introduction:
Haworth formula indicates the connectivity of atoms in a ring structure. The linear structure should be used in the conversion.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- What products are obtained from the reduction of a. d-idose? b. d-sorbose?arrow_forwardHow many stereoisomers are possible for a. a ketoheptose? b. an aldoheptose? c. a ketotriose?arrow_forwarda.Locate the stereogenic centers in the ball-and-stick model of ezetimibe (trade name Zetia), a cholesterol-lowering drug. b. Label each stereogenic center as R or S.arrow_forward
- Labeling a Stereogenic Center as R or S Label the stereogenic center in each compound as R or S.arrow_forwardKk.305.arrow_forwardFor D-arabinose:a. Draw its enantiomer.b. Draw an epimer at C3.c. Draw a diastereomer that is not an epimer.d. Draw a constitutional isomer that still contains a carbonyl group.arrow_forward
- Label the stereogenic center in attached compound as R or S.arrow_forwardFor D-lyxose i. Draw an epimer at C3. ii. Draw a diastereomer that is not an epimer. iii. Draw the anomers of -D-lyxofuranose and -D-lyxopyranose. iv. Draw the product as Haworth projection when reacted with methanol catalysed by acid.arrow_forwarda. How many stereogenic centers are present in α-D-galactose? b. Label the hemiacetal carbon in α-D-galactose. c. Draw the structure of β-D-galactose. d. Draw the structure of the polyhydroxy aldehyde that cyclizes to α- and β-D-galactose.arrow_forward
- Label each stereogenic center as R or S.arrow_forwardDraw the products formed when β-D-galactose is treated with each reagent. a. Ag2O + CH3I b. NaH + C6H5CH2Cl c. The product in (b), then H3O+ d. Ac2O + pyridine e. C6H5COCl + pyridine f. The product in (c), then C6H5COCl + pyridinearrow_forwardPenicillin is well-known anti-bacterial agent discovered by Alexander Fleming. A O. N- H. В Which box contains a carbocyclic unsaturation? IZarrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

