
Concept explainers
Interpretation:
The acetals in sucralose should be located and each
Concept Introduction:
Addition of one molecule of alcohol to an
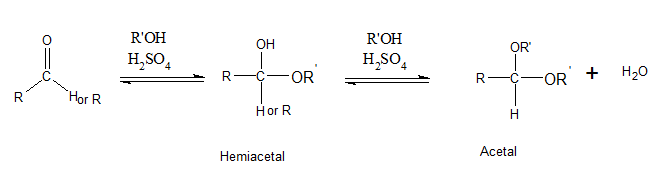
likewise, a disaccharide is formed when a hemiacetal of a one monosaccharide is reacted with a hydroxyl group of a second monosaccharide forming an acetal.
Primary alkyl halide are halides that have one alkyl group bonded to the same carbon as the halide. Likewise, secondary and tertiary halides are halides that have two and three alkyl groups bonded to same carbon as halide respectively.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Fructose, present with glucose in honey, reacts with Benedicts reagent. Circle the structural features that enable fructose to react.arrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forwardDraw the products formed when each acetal is treated with aqueous acid.arrow_forward
- Draw the product formed in each reaction.arrow_forwardDraw the products formed when attached acetal is treated with aqueous acid ?arrow_forwardDraw the major products of the SN1 reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, where applicable. Ignore any inorganic byproducts. H OH HCI Q Please select a drawing or reagent from the question areaarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
