
Organic Chemistry, Loose-leaf Version
8th Edition
ISBN: 9781305865549
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 21.28P
Propose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.
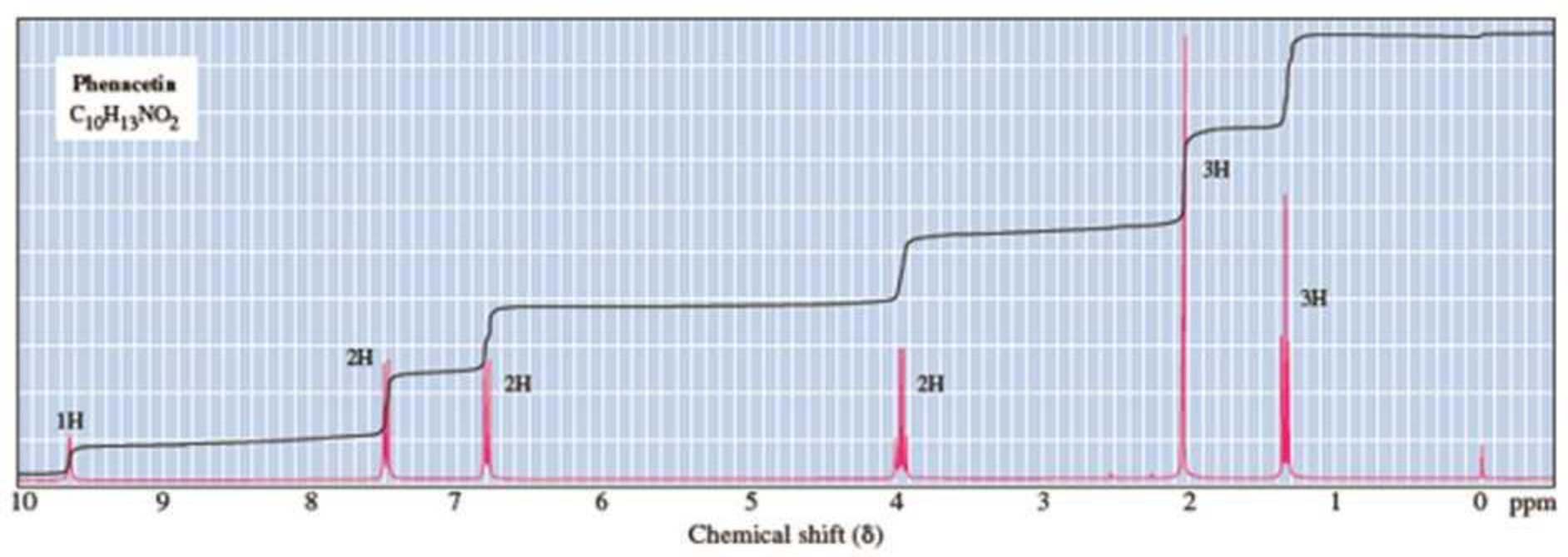
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The 1H-NMR spectrum of compound B,C7H14O , consists of the following signals: δ 0.9 (t, 6H), 1.6 (sextet, 4H), and 2.4 (t, 4H).
Draw the structural formula of compound B.
There are four esters with molecular formula C4H8O2. How can they be distinguished by 1H NMR?
Draw the H1 NMR spectra of ethylcyclopropane. Draw the chemical structure and predict the proton splitting and chemical shifts.
Chapter 21 Solutions
Organic Chemistry, Loose-leaf Version
Ch. 21.2 - Construct a Frost circle for a planar...Ch. 21.2 - Which compound gives a signal in the 1H-NMR...Ch. 21.2 - Prob. 21.3PCh. 21.2 - Prob. 21.4PCh. 21.3 - Prob. 21.5PCh. 21.4 - Arrange these compounds in order of increasing...Ch. 21.4 - Prob. AQCh. 21.4 - Prob. BQCh. 21.4 - Prob. CQCh. 21.5 - Prob. 21.7P
Ch. 21 - Name the following compounds and ions.Ch. 21 - Prob. 21.9PCh. 21 - Draw a structural formula for each compound. (a)...Ch. 21 - Molecules of 6,6-dinitrobiphenyl-2,2-dicarboxylic...Ch. 21 - Following each name is the number of Kekul...Ch. 21 - Prob. 21.13PCh. 21 - Prob. 21.14PCh. 21 - Prob. 21.15PCh. 21 - Which of the molecules and ions given in Problem...Ch. 21 - Prob. 21.17PCh. 21 - Naphthalene and azulene are constitutional isomers...Ch. 21 - Prob. 21.19PCh. 21 - Prob. 21.20PCh. 21 - Following are IR and 1H-NMR spectra of compound D....Ch. 21 - Compound E (C8H10O2) is a neutral solid. Its mass...Ch. 21 - Following are 1H-NMR and 13C-NMR spectral data for...Ch. 21 - Following are 1H-NMR and 13C-NMR spectral data for...Ch. 21 - Compound H (C8H6O3) gives a precipitate when...Ch. 21 - Compound I (C11H14O2) is insoluble in water,...Ch. 21 - Propose a structural formula for compound J...Ch. 21 - Propose a structural formula for the analgesic...Ch. 21 - Prob. 21.29PCh. 21 - Prob. 21.30PCh. 21 - Given here are 1H-NMR and 13C-NMR spectral data...Ch. 21 - Prob. 21.32PCh. 21 - Prob. 21.33PCh. 21 - Prob. 21.34PCh. 21 - Arrange the molecules and ions in each set in...Ch. 21 - Prob. 21.36PCh. 21 - Prob. 21.37PCh. 21 - From each pair, select the stronger base.Ch. 21 - Prob. 21.39PCh. 21 - Prob. 21.40PCh. 21 - Prob. 21.41PCh. 21 - Prob. 21.42PCh. 21 - Following is an equation for iodination of...Ch. 21 - Prob. 21.44PCh. 21 - Prob. 21.45PCh. 21 - Prob. 21.46PCh. 21 - When warmed in dilute sulfuric acid,...Ch. 21 - In the chemical synthesis of DNA and RNA, hydroxyl...Ch. 21 - Prob. 21.49PCh. 21 - Write the products of the following sequences of...Ch. 21 - Prob. 21.51PCh. 21 - Show how to convert 1-phenylpropane into the...Ch. 21 - Prob. 21.53PCh. 21 - Cromolyn sodium, developed in the 1960s, has been...Ch. 21 - Prob. 21.55PCh. 21 - Prob. 21.56PCh. 21 - Prob. 21.57PCh. 21 - Prob. 21.58PCh. 21 - Prob. 21.59PCh. 21 - Prob. 21.60PCh. 21 - Prob. 21.61PCh. 21 - Prob. 21.62PCh. 21 - Prob. 21.63PCh. 21 - Following is a synthesis for toremifene, a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forwardCompound 2 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Provide structure for compound 2, explain how you reached your conclusion.arrow_forward16-17 Propylamine (bp 48°C), ethylmethylamine (bp 37°C), and trimethylamine (bp 3°C) are constitutional isomers with the molecular formula C3HgN. Account for the fact that trimethylamine has the lowest boiling point of the three and propylamine has the highest.arrow_forward
- A ¹H NMR spectrum is shown for a molecule with the molecular formula of C9H10O2. Draw the structure that best fits this data. 1H 11 10 9 8 SH 7 4 214 21 1 pom Qarrow_forwardThe 1H NMR spectrum of 2-chloroacetamide (ClCH2CONH2) shows three signals at 4.02, 7.35, and 7.60 ppm. What protons give rise to each signal? Explain why three signals are observed.arrow_forward11 10 9 8 7 9 +6 5 -3 2 1 ppmarrow_forward
- Ciprofloxacin is a member of the fluoroquinolone class of antibiotics.(a) Which of its rings are aromatic?(b) Which nitrogen atoms are basic?(c) Which protons would you expect to appear between d 6 and d 8 in the proton NMR spectrum?arrow_forwardAn amine of unknown structure contains one nitrogen and nine carbon atoms. The 13C-NMR spectrum shows only five signals, all between 20 and 60 ppm. Three cycles of Hofmann elimination sequence [(1) CH3I; (2) Ag2O, H3O; (3) heat] give trimethylamine and 1,4,8-nonatriene. Propose a structural formula for the amine.arrow_forwardAn NMR spectrum was run for the following compound. Assign proton NMR peaks and explain.arrow_forward
- Consider the ¹H NMR spectrum of the following compound, and rank the signals in terms of increasing chemical shift. Identify the proton(s) giving rise to each signal: Cl H OHarrow_forwardCompound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY