
(a)
Interpretation:
The major product of the reaction between propionyl chloride and
Concept introduction:
Acid chloride is a acid of
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride and
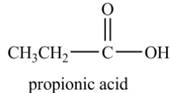
Explanation of Solution
Propionyl chloride undergoes hydrolysis reaction to from propionic acid. The water molecule acts as a nucleophile and attacks on the carbonyl carbon atom of propionyl chloride. The resultant compound lose
The corresponding
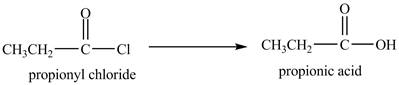
Figure 1
The major product of the reaction between propionyl chloride and
(b)
Interpretation:
The major product of the reaction between propionyl chloride, ethanethiol, and pyridine, at
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride, ethanethiol, and pyridine, at
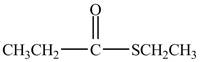
Explanation of Solution
Propionyl chloride is a acyl chloride. It has a electrophilic carbonyl carbonyl carbon atom. The ethanethiol molecule acts as a nucleophile and attacks on the carbonyl carbon atom of propionyl chloride. The resultant compound lose
The corresponding chemical reaction is shown below.

Figure 2
The major product of the reaction between propionyl chloride, ethanethiol, and pyridine, at
(c)
Interpretation:
The major product of the reaction between propionyl chloride,
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride,
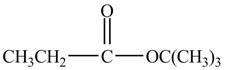
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. The compound
The corresponding chemical reaction is shown below.

Figure 3
The major product of the reaction between propionyl chloride,
(d)
Interpretation:
The major product of the reaction between propionyl chloride and
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride and
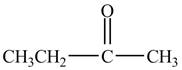
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. The compound
The corresponding chemical reaction is shown below.
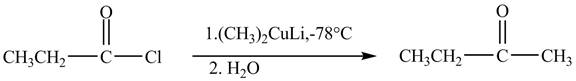
Figure 4
The major product of the reaction between propionyl chloride and
(e)
Interpretation:
The major product of the reaction between propionyl chloride and ![]() sulfur poison) is to be predicated.
sulfur poison) is to be predicated.
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride and ![]() sulfur poison) is shown below.
sulfur poison) is shown below.
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. propionyl chloride reduces similary as carbonylic acid with ![]() sulfur poison to an
sulfur poison to an
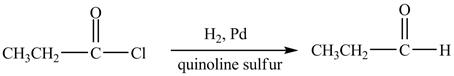
Figure 5
The major product of the reaction between propionyl chloride and ![]() sulfur poison) is shown in Figure 5
sulfur poison) is shown in Figure 5
(f)
Interpretation:
The major product of the reaction between propionyl chloride and
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride and
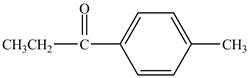
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. The compound undergoes Fredal craft acylation reaction with toluene in the presence of catalyst
The corresponding chemical reaction is shown below.
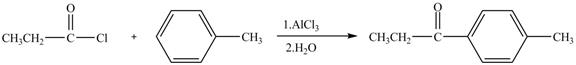
Figure 6
The major product of the reaction between propionyl chloride and
(g)
Interpretation:
The major product of the reaction between propionyl chloride and
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride and
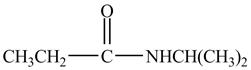
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. The compound
The corresponding chemical reaction is shown below.
![]()
Figure 7
The major product of the reaction between propionyl chloride and
(h)
Interpretation:
The major product of the reaction between propionyl chloride and sodium benzoate is to be predicated.
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride and sodium benzoate is shown below.
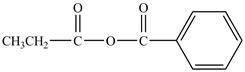
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. Th The compound sodium benzoate reacts with propionyl chloride. The resultant compound lose
The corresponding chemical reaction is shown below.
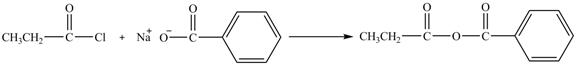
Figure 8
The major product of the reaction between propionyl chloride and sodium benzoate is shown in Figure 8.
(i)
Interpretation:
The major product of the reaction between propionylchloride,
Concept introduction:
Acid chloride is a type of carboxylic acid derivatives. The general formula of acid chloride is
Answer to Problem 21.33AP
The major product of the reaction between propionyl chloride,
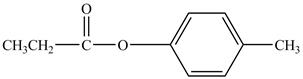
Explanation of Solution
Propionyl chloride is an acyl chloride. It has an electrophilic carbonyl carbon atom. Th The compound
The corresponding chemical reaction is shown below.

Figure 9
The major product of the reaction between propionyl chloride,
Want to see more full solutions like this?
Chapter 21 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- The odor of ripe bananas and many other fruits is due to the presence of esters. For example: Banana oil (isopentyl acetate) (a) Write the name (common or IUPAC) of the ester responsible for the fragrance of the following: pineapple, orange, apple, peach, & lavender (b) Choose one fragrant from (a) and name the alcohol and the carboxylic acid needed to synthesize this ester. (c) Show the detailed mechanism of the Fischer Esterification reaction that will be involved in the synthesis of the fragrant you have chosen in part (a).arrow_forwardWrite a structural formula for each of the following compounds: (a) m-Chlorobenzoyl chloride (b) Trifluoroacetic anhydride (c) cis-1,2-Cyclopropanedicarboxylic anhydride (d) Ethyl cycloheptanecarboxylate (e) 1-Phenylethyl acetate (f) 2-Phenylethyl acetate (g) p-Ethylbenzamide (h) N-Ethylbenzamide (i) 2-Methylhexanenitrilearrow_forwardDraw the structural formulas of the following compounds:(a) 2,3-Dimethylpentanal(b) 1,3-Dibromopropanone(c) 4-hydroxy-4-methylhexan-2-onearrow_forward
- 6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forwardDraw structural formulas for these ketones. (a) Ethyl isopropyl ketone (b) 2-Chlorocyclohexanone (c) 2,4-Dimethyl-3-pentanone (d) Diisopropyl ketone (e) Acetone (f) 2,5-Dimethylcyclohexanonearrow_forwardDimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of female hamsters, acts as a sexual attractant for the male hamster. Write an equation for its synthesis from methanethiol.arrow_forward
- (b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forwardThe hydrocarbon fluorene was treated with potassium t-butoxide in an acid-base reaction, giving the fluorenide anion and t-butyl alcohol. (a) Which way does the equilibrium lie, and by how much? b) What is the proportion of the fluorenide anion to fluorene? (c) Why is fluorene so highly acidic, considering the pKa of an average alkane is above 50?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





