
ALEKS 360 CHEMISTRY ACCESS
4th Edition
ISBN: 9781264104369
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 21.33P
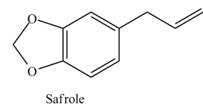
Safrole is a naturally occurring acetal isolated from sassafras plants. Once used as a common food additive in root beer and other beverages, it is now banned because it is carcinogenic. What compounds are formed when safrole is hydrolyzed with aqueous acid?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.
Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)
Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.
Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+):
CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)
a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield?
b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield?
c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?
Chapter 21 Solutions
ALEKS 360 CHEMISTRY ACCESS
Ch. 21 - Rank the following compounds in order of...Ch. 21 - Prob. 21.2PCh. 21 - Give the IUPAC name for each aldehyde.Ch. 21 - Prob. 21.4PCh. 21 - Give the IUPAC name for each ketone.Ch. 21 - Prob. 21.6PCh. 21 - Prob. 21.7PCh. 21 - The boiling point of is significantly higher than...Ch. 21 - Which carbonyl group in each pair absorbs at a...Ch. 21 - Problem 21.10 Draw the structure of all...
Ch. 21 - Prob. 21.11PCh. 21 - Prob. 21.12PCh. 21 - Prob. 21.13PCh. 21 - Prob. 21.14PCh. 21 - Problem 21.15 Draw the product of each...Ch. 21 - Prob. 21.16PCh. 21 - Prob. 21.17PCh. 21 - Outline a synthesis of each Wittig reagent from...Ch. 21 - Draw the products including stereoisomers formed...Ch. 21 - Prob. 21.20PCh. 21 - Prob. 21.21PCh. 21 - Show two methods to synthesize each alkene: a...Ch. 21 - Problem 21.22 The product formed when reacts with...Ch. 21 - What 1 amine and carbonyl compound are needed to...Ch. 21 - Prob. 21.25PCh. 21 - Prob. 21.26PCh. 21 - Prob. 21.27PCh. 21 - Problem 21.28 Draw a stepwise mechanism for the...Ch. 21 - Problem 21.29 Draw the products of each...Ch. 21 - Problem 21.30 Label each compound as an acetal, a...Ch. 21 - Problem 21.31 Draw a stepwise mechanism for the...Ch. 21 - Problem 21.32 Draw the products of each...Ch. 21 - Problem 21.33 Safrole is a naturally occurring...Ch. 21 - Problem 21.35 How would you use a protecting group...Ch. 21 - Prob. 21.35PCh. 21 - Problem 21.37 Two naturally occurring compounds...Ch. 21 - Problem 21.38 Draw the products of each...Ch. 21 - Prob. 21.38PCh. 21 - Problem 21.40 (a) Give the IUPAC name for A and B....Ch. 21 - 21.41 Rank the following compounds in order of...Ch. 21 - Prob. 21.41PCh. 21 - Give the IUPAC name for each compound. a. d. g. b....Ch. 21 - Give the structure corresponding to each name. a....Ch. 21 - Including stereoisomers, draw the 11 aldehydes...Ch. 21 - Prob. 21.45PCh. 21 - Prob. 21.46PCh. 21 - What alkyl halide is needed to prepare each Wittig...Ch. 21 - 21.46 Draw the products of each reaction.
a. e....Ch. 21 - Prob. 21.49PCh. 21 - Prob. 21.50PCh. 21 - Prob. 21.51PCh. 21 - Prob. 21.52PCh. 21 - Prob. 21.53PCh. 21 - Rank the following compounds in order of...Ch. 21 - Which compound forms the higher concentration of...Ch. 21 - Prob. 21.56PCh. 21 - What Wittig reagent and carbonyl compound are...Ch. 21 - Prob. 21.58PCh. 21 - What reagents are needed to convert each compound...Ch. 21 - What reagents are needed to convert each compound...Ch. 21 - Prob. 21.61PCh. 21 - Devise a synthesis of each alkene using a Wittig...Ch. 21 - Devise a synthesis of each compound from...Ch. 21 - Prob. 21.64PCh. 21 - Prob. 21.65PCh. 21 - Prob. 21.66PCh. 21 - Prob. 21.67PCh. 21 - Prob. 21.68PCh. 21 - Prob. 21.69PCh. 21 - 21.64 Draw a stepwise mechanism for the following...Ch. 21 - 21.65 Draw a stepwise mechanism f or the following...Ch. 21 - Prob. 21.72PCh. 21 - 21.67 Draw a stepwise mechanism for each...Ch. 21 - 21.68 Draw a stepwise mechanism for the following...Ch. 21 - Prob. 21.75PCh. 21 - Prob. 21.76PCh. 21 - Prob. 21.77PCh. 21 - Prob. 21.78PCh. 21 - Prob. 21.79PCh. 21 - 21.73 Although the carbonyl absorption of cyclic...Ch. 21 - 21.74 Use the and data to determine the...Ch. 21 - 21.75 A solution of acetone in ethanol in the...Ch. 21 - Compounds A and B have molecular formula ....Ch. 21 - 21.77 An unknown compound C of molecular formula ...Ch. 21 - 21.78 An unknown compound D exhibits a strong...Ch. 21 - Prob. 21.86PCh. 21 - -D-Glucose, a hemiacetal, can be converted to a...Ch. 21 - 21.81 Draw a stepwise mechanism for the following...Ch. 21 - Prob. 21.89PCh. 21 - Prob. 21.90PCh. 21 - Prob. 21.91PCh. 21 - 21.86 Draw stepwise mechanism for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Fenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid (CH3CO2H)?arrow_forwardWhat products are formed when each compound is treated with aqueous acid?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Part A Given 7.30 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. Part B A chemist ran the reaction and obtained 5.95 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 7.30 gg of…arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.45 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. A chemist ran the reaction and obtained 5.50 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 8.45 gg of butanoic acid and excess…arrow_forwardEtoposide, sold as a phosphate derivative with the trade name ofEtopophos, is used for the treatment of lung cancer, testicular cancer,and lymphomas. What products are formed when all of the acetals are hydrolyzed with aqueous acid?arrow_forward
- Amino acids such as glycine are the building blocks of large molecules called proteins that give structure to muscle, tendon, hair, and nails. What product is formed when glycine is treated with concentrated HCl? What product is formed when glycine is treated with NaOH?arrow_forwardWhat product is formed when a solution of A to B is treated with mild base? This reaction is the first step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forwardGive the IUPAC name (including any E,Z designation) for each unsaturated aldehyde. Neral is obtained from lemon grass, and cucumber aldehyde contributes to the aroma of a fresh mango.arrow_forward
- Benzaldehyde is the compound principally responsible for the odor of almonds. What product is formed when benzaldehyde is treated with H2, Pd?arrow_forwardAspirin is an anti-inflammatory agent because it inhibits the conversion of arachidonic acid to prostaglandins by the transfer of its acetyl group (CH3CO−) to an OH group at the active site of an enzyme. This reaction, called transesterification, results in the conversion of one ester to another by a nucleophilic acyl substitution reaction. Draw a stepwise mechanism for the given transesterification.arrow_forwardWhat products are formed when attached acetal is hydrolyzed with aqueousacid?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY