
(a)
Interpretation:
The mechanism and the major organic product for the given reaction are to be drawn.
Concept introduction:
Migratory Aptitude in a Baeyer–Villiger Oxidation:
Answer to Problem 21.60P
The mechanism and the major organic product for the given reaction are:
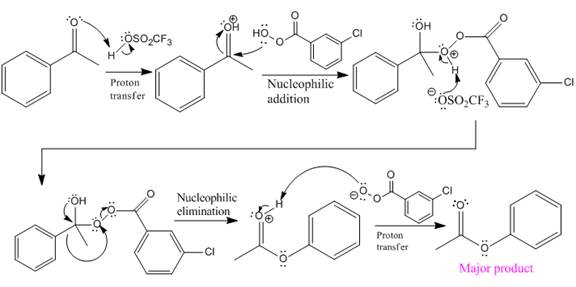
Explanation of Solution
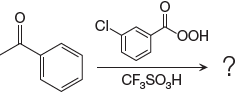
The given reaction is:
The ketone, substrate in the reaction, is asymmetrical ketone. carbonyl C is bonded to a primary alkyl group and an aryl group. According to migratory aptitude, the aryl group has greater migratory aptitude, so its bond will prefentially break.
In this reaction, an O atom from the acid is inserted between carbonyl C and phenyl group, initially bonded to the carbonyl C. C=O is activated by
The complete mechanism and the ester formed as a product for the reaction are:
The mechanism and the product for the reaction are drawn on the basis of the given reaction conditions.
(b)
Interpretation:
The mechanism and the major organic product for the given reaction are to be drawn.
Concept introduction:
Migratory Aptitude in a Baeyer–Villiger Oxidation:
Answer to Problem 21.60P
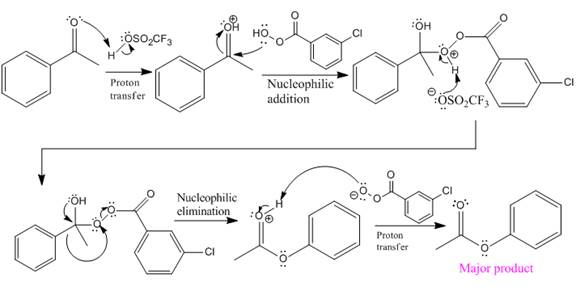
The mechanism and the major organic product for the given reaction are:
Explanation of Solution
The given reaction is:
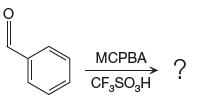
The ketone, substrate in the reaction, is asymmetrical ketone. carbonyl C is bonded to a H atom and an aryl group. According to the migratory aptitude, the H atom has greater migratory aptitude, so its bond will prefentially break.
In this reaction, an O atom from the acid is inserted between carbonyl C and the H atom, initially bonded to carbonyl C. C=O is activated by
The complete mechanism and the ester formed as a product for the reaction are:
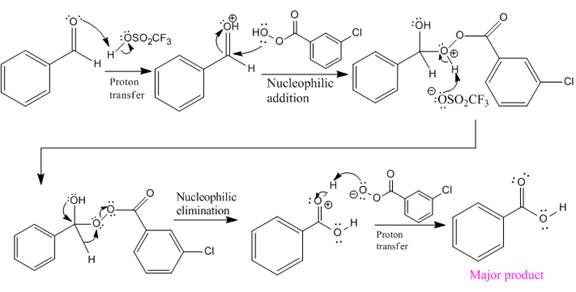
The mechanism and the product for the reaction are drawn on the basis of given reaction conditions.
(c)
Interpretation:
The mechanism and the major organic product for the given reaction are to be drawn.
Concept introduction:
Migratory Aptitude in a Baeyer–Villiger Oxidation:
Answer to Problem 21.60P
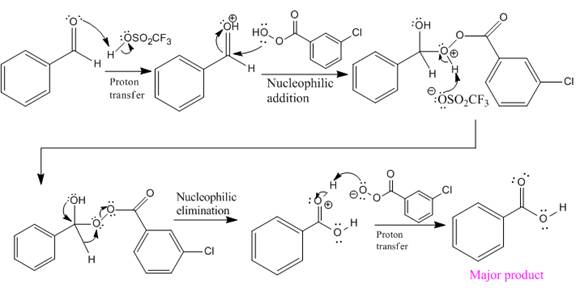
The mechanism and the major organic product for the given reaction are:
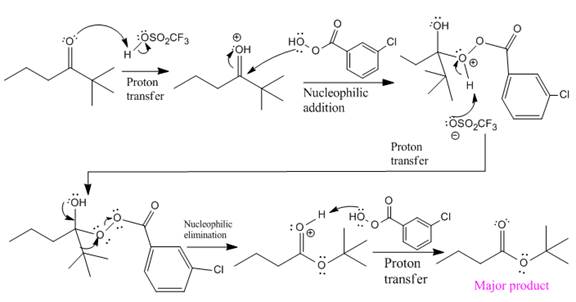
Explanation of Solution
The given reaction is:
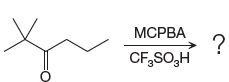
The ketone, substrate in the reaction, is asymmetrical ketone. carbonyl C is bonded to a primary alkyl group and tertiary alkyl group. According to migratory aptitude, the tertiary alkyl group has greater migratory aptitude, so its bond will prefentially break.
In this reaction an O atom from the acid is inserted between carbonyl C and the tertiary alkyl group, initially bonded to carbonyl C. C=O is activated by
The complete mechanism and the ester formed as a product for the reaction are:

The mechanism and the product for the reaction are drawn on the basis of given reaction conditions.
Want to see more full solutions like this?
Chapter 21 Solutions
Get Ready for Organic Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





