
Concept explainers
Use the
Compound A Molecular formula:
Compound B Molecular formula:
Compound C Molecular formula:
Compound D Molecular formula:
Interpretation: The structure of each compound from the given
Concept introduction: Spectroscopy method is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton
Answer to Problem 21.74P
The structure of A compound from the given data of
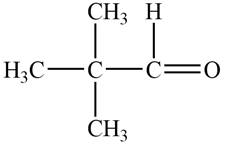
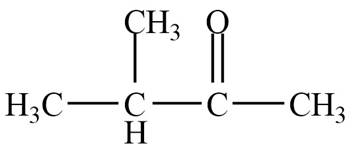
The structure of B compound from the given data of
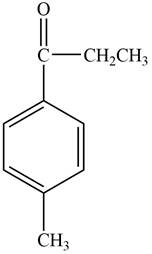
The structure of C compound from the given data of
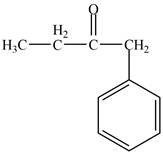
The structure of D compound from the given data of
Explanation of Solution
Structure determination of Compound A:
The molecular formula of the Compound A is
Information from
The
The
Information from
The observed chemical shift value at
The observed chemical shift value at
Thus, the structure of Compound A from the given
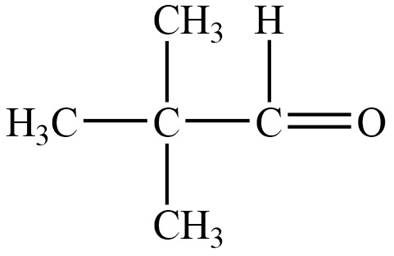
Figure 1
Structure determination of Compound B:
The molecular formula of the Compound B is
Information from
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
Thus, the structure of Compound B from the given
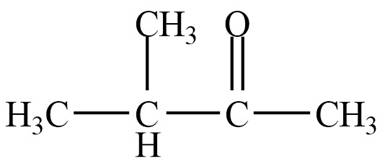
Figure 2
Structure determination of Compound C:
The molecular formula of the Compound C is
Information from
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift values at
Thus, the structure of Compound C from the given
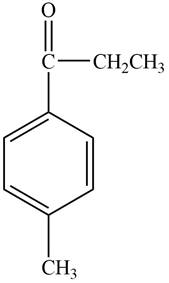
Figure 3
Structure determination of Compound D:
The molecular formula of the Compound D is
Information from
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
Thus, the structure of Compound D from the given
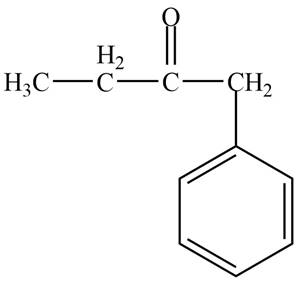
Figure 4
The structures of A, B, C, and D compounds from the given data of
Want to see more full solutions like this?
Chapter 21 Solutions
ORGANIC CHEMISTRY LL W/SSM+CONNECT+KIT
- Assume a compound with the formula C4H8O. a) How many double bonds and/or rings does your compound contain? b) If your compound shows an infrared absorption peak at 1715 cm-1, what functional group does it have? c) If your compound shows a single 1H NMR absorption peak at 2.1 δ, what is its structure?arrow_forwardDeduce the structures of compounds A and B, two of the major components of jasmine oil, from the given data. Compound A: C9H10O2; IR absorptions at 3091–2895 and 1743 cm-1; 1H NMR signals at 2.06 (singlet, 3 H), 5.08 (singlet, 2 H), and 7.33 (broad singlet, 5 H) ppm. Compound B: C14H12O2; IR absorptions at 3091–2953 and 1718 cm-1; 1H NMR signals at 5.35 (singlet, 2 H) and 7.26–8.15 (multiplets, 10 H) ppm.arrow_forwardCompound C has the molecular formula of C6H12O2 with the following IR data. The mass and NMR (1H and 13C NMR) spectra are shown below. IR (cm-1): 2800, 2950, 1710 and 1120 cm-1arrow_forward
- 19. A compound with molecular formula C6H12O2 exhibits two singlets in its 1H NMR spectrum, at d 1.4 (9H) and d 2.0 (3H). Its IR spectrum shows a strong absorption band near 1740 cm-1. What is the structure for this compound? Show the correlation of the spectra with the molecular structure.arrow_forward15. Compound C has a molecular weight of 94.54 and the 1H-NMR spectrum shows four signals - a triplet at 3.81 ppm, a triplet at 3.63 ppm, a singlet at 2.19 ppm, and a triplet of triplets at 2.02 ppm. The mass, IR, and 13C-NMR spectra of compound C are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Identify C and explain your reasoning. refer to picturearrow_forwardA compound of formula C6H10O2 shows only two absorptions in the proton NMR: a singlet at 2.67 ppm and a singlet at2.15 ppm. These absorptions have areas in the ratio 2:3. The IR spectrum shows a strong absorption at 1708 cm-1. Proposea structure for this compound.arrow_forward
- Following are IR and 1H-NMR spectra of compound D. The mass spectrum of compound D shows a molecular ion peak at m/z 136, a base peak at m/z 107, and other prominent peaks at m/z 118 and 59. Q.) Propose structural formulas for ions in the mass spectrum at m/z 118, 107, and 59.arrow_forwardPropose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm−1 and the following 1H NMR data:arrow_forward
