
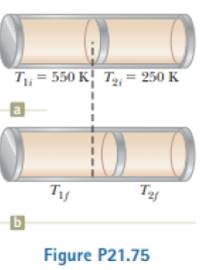
A cylinder is closed at both ends and has insulating EZZ3 walls. It is divided into two compartments by an insulating piston that is perpendicular to the axis of the cylinder as shown in Figure P21.75a. Each compartment contains 1.00 mol of oxygen that behaves as an ideal gas with γ = 1.40. Initially, the two compartments haw equal volumes and their temperatures are 550 K and 250 K. The piston is then allowed to move slowly parallel to the axis of the cylinder until it comes to rest at an equilibrium position (Fig. P2l.75b). Find the final temperatures in the two compartments.
Trending nowThis is a popular solution!

Chapter 21 Solutions
Bundle: Physics for Scientists and Engineers, Technology Update, 9th Loose-leaf Version + WebAssign Printed Access Card, Multi-Term
- In Figure P19.22, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? Figure P19.22 (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D?arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally, (f) Find Q, W, and Eint for each of the processes, (g) For the whole cycle A B C A, find Q, W, and Eint.arrow_forwardIn Figure P17.32, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D? Figure P17.32arrow_forward
- (a) An ideal gas occupies a volume of 1.0 cm3 at 20.C and atmospheric pressure. Determine the number of molecules of gas in the container, (b) If the pressure of the 1.0-cm3 volume is reduced to 1.0 1011 Pa (an extremely good vacuum) while the temperature remains constant, how many moles of gas remain in the container?arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forwardCase Study When a constant-volume thermometer is in thermal contact with a substance whose temperature is lower than the triple point of water, how does the right tube in Figure 19.22 need to be moved? Explain. FIGURE 19.22 1 Gas in the constant-volume gas thermometer is at Ti, and the mercury in the manometer is at height hi above the gasmercury boundary. 2 The thermometer is placed in thermal contact with an object, and its temperature increases. The increased temperature increases the gas volume. 3 By raising the right-hand tube of the mercury manometer, the gas volume is restored to its original size. The mercury is now at hi + h above the gasmercury boundary. This increase in height is a result of the increase in gas temperature and pressure.arrow_forward
- A bicycle tire has a pressure of P1 = 6.95 × 105 Pa at a temperature of T = 19.5°C and contains V = 2.00 L of gas. a. You open the valve on the bicycle tire and let out an amount of air which has a volume Va at atmospheric pressure Pa and at the temperature T of the tire. How many moles will be in this amount of air, in terms of variables given in the problem statement? b. What will the pressure of the tire be now, in terms of the variables given in the problem statement? Assume the tire temperature and volume remain constant. c. What will this pressure be, in pascals, if the amount of air let out was 110 cm3?arrow_forwardA tank contains one mole of nitrogen gas at a pressure of 5.20 atm and a temperature of 24.5°C. The tank (which has a fixed volume) is heated until the pressure inside triples. What is the final temperature of the gas? °C (b)A cylinder with a moveable piston contains one mole of nitrogen, again at a pressure of 5.20 atm and a temperature of 24.5°C. Now, the cylinder is heated so that both the pressure inside and the volume of the cylinder double. What is the final temperature of the gas? °Carrow_forwardA hollow container is filled with an ideal gas. the container is designed to maintain a constant pressure by allowing gas to enter or leave as needed. at all times the gas and container are in thermal equilibrium. initially the temperature is 560 K. then the temperature increases to 2688 K and the volume increases by a factor of 1.12. determine the coefficient of volume expansion for the container, and the ratio of the final number of moles to initial moles.arrow_forward
- A hollow container is filled with an ideal gas. The container is designed to maintain a constant pressure by allowing gas to enter or leave as needed. At all times the gas & container are in thermal equilibrium. Initially the temperature is 2350 K. Then the temperature decreases to 500 K and the volume decreases to 0.81 times the initial volume. Determine the coefficient of volume expansion for the container and the ratio of the final number of moles to the initial number of moles. Bcontainer nf ni = = K-1arrow_forward4.0 moles of krypton gas are in a 0.20m3 container. The pressure is 1.663 x 105 Pa. a, What is the temperature (to the nearest K)? The volume contracts to 0.10m3. The pressure is held constant. b, How much work was done by the gas during the volume contraction? c, What is the temperature after the volume contraction (to the nearest K)?arrow_forward(a) A tank contains one mole of helium gas at a pressure of 6.35 atm and a temperature of 22.0°C. The tank (which has a fixed volume) is heated until the pressure inside triples. What is the final temperature of the gas? °C (b) A cylinder with a moveable piston contains one mole of helium, again at a pressure of 6.35 atm and a temperature of 22.0°C. Now, the cylinder is heated so that both the pressure inside and the volume of the cylinder double. What is the final temperature of the gas? °Carrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





