
Concept explainers
Draw enol tautomer(s) for each compound. Ignore stereoisomers.
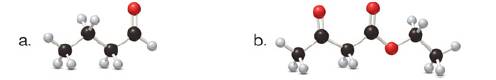
(a)
Interpretation: Enol tautomer(s) of the given compound is to be drawn.
Concept introduction: Tautomers are the isomers which differ only in the position of the hydrogens and electrons of electronegative element, generally oxygen. There is no change in the carbon skeleton of the compound. This phenomenon which involves simple proton transfer in an intramolecular fashion is known as tautomerism.
The very common example of tautomerism is Keto-enol tautomerism. It can be acid or base catalysed.
Answer to Problem 30P
. The enol tautomer of this compound is shown below:
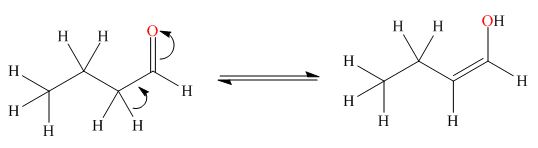
Explanation of Solution
Tautomers are isomers which differ only in the position of the protons and electrons of the compound. There is no change in the carbon skeleton of the compound. The ball and stick model as shown in Figure 1.
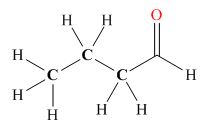
Figure 1
The enol tautomer of this compound is shown in Figure 2.

Figure 2
The tautomer of the given compound is showed in Figure 2.
(b)
Interpretation: Enol tautomer(s) of the given compound is to be drawn.
Concept introduction:
Tautomers are the isomers which differ only in the position of the hydrogens and electrons of electronegative element, generally oxygen. There is no change in the carbon skeleton of the compound. This phenomenon which involves simple proton transfer in an intramolecular fashion is known as tautomerism.
The very common example of tautomerism is Keto-enol tautomerism. It can be acid or base catalysed.
Answer to Problem 30P
. The enol tautomer of this compound is shown below:
Explanation of Solution
Tautomers are isomers which differ only in the position of the protons and electrons of the compound. There is no change in the carbon skeleton of the compound. The ball and stick model as shown in Figure 3.
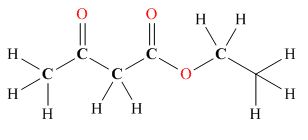
Figure 3
The enol tautomer of this compound is shown in Figure 4.

Figure 4
The tautomer of the given compound is showed in Figure 4.
Want to see more full solutions like this?
Chapter 21 Solutions
LL ORG CHEM
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. CH3COCl, AlCl3arrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. Part (b), then CH3COCl, AlCl3arrow_forwardMatch each reagent to the product that it forms. Multiple reagents may form the same product. нох Reagent Reagents SOCI2, pyridine: C CISO2CH3, pyridine: E HCI: A PCI 3: A A) B) "It "ft "bl H₂O D) E) F) پہلے علی علیہarrow_forward
- Synthesize each compound from cyclohexanol using any other organic or inorganic compounds. CH,OH a. g. (Each cyclohexane ring must come from cyclohexanol.) COOH b. d. h. сно CHs (Each cyclohexane ring must come from cyclohexanol.)arrow_forwardDevise a synthesis of each substituted cyclopropane. Use acetylene (HC≡CH) as a starting material in part (a), and cyclohexanone as a starting material in part (b). You may use any other organic compounds and any needed reagents.arrow_forwardDraw a stepwise mechanism for the following reaction. .COOH H2SO4 но H20arrow_forward
- Answer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.a.In Chapter 11, we learned that most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols. b.Explain why the enol O—H proton is more acidic than an alcohol O—H proton. c. Why is curcumin colored? d.Explain why curcumin is an antioxidant.arrow_forwardDraw the organic products formed when attached allylic alcohol A is treated with following reagent. [1] PBr3; [2] LiAlH4; [3] H2Oarrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. NaNO2, HClarrow_forward
- (a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C=C and an OH group. 2-methylcyclohexanonearrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] CH3C≡CLi; [2] H2Oarrow_forwardDraw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster than chlorobenzene in this type of reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
