
EBK ORGANIC CHEMISTRY
12th Edition
ISBN: 9781119233664
Author: Snyder
Publisher: VST
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 21, Problem 5PP
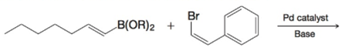
PRACTICE PROBLEM 21.5
What is the product of the following Suzuki-Miyaura coupling?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Give reasons :(a) n-Butyl bromide has higher boiling point than f-butyl bromide.(b) Racemic mixture is optically inactive.(c) The presence of nitro group (-NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
Account for the following:(i) Aniline does not give Friedel-Crafts reaction.(ii) Ethylamine is soluble in water whereas aniline is not.(iii) pKb of methylamine is less than that of aniline.
A compound A, C7H12, was found to be optically active. On catalytic reduction over platinum catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, C7H16. On ozonolysis of A, two fragments were obtained. One fragment was identified as acetic acid. The other fragment, compound C, was an optically active carboxylic acid, C5H10O2. Write the reactions and draw structures for A, B and C.
Chapter 21 Solutions
EBK ORGANIC CHEMISTRY
Ch. 21 - PRACTICE PROBLEM 21.1
For each of the following...Ch. 21 - Prob. 2PPCh. 21 - PRACTICE PROBLEM 21.3 What product would you...Ch. 21 - Prob. 4PPCh. 21 - PRACTICE PROBLEM 21.5 What is the product of the...Ch. 21 - Prob. 6PPCh. 21 - Prob. 7PPCh. 21 - Prob. 8PPCh. 21 - Prob. 9PPCh. 21 - Prob. 10PP
Ch. 21 - Prob. 11PPCh. 21 - Practice Problem 21.12 What products would form...Ch. 21 - Prob. 13PCh. 21 - Prob. 14PCh. 21 - How would you synthesize each of the following...Ch. 21 - Prob. 16PCh. 21 - Predict the product(s) for each of the following...Ch. 21 - Prob. 18PCh. 21 - Prob. 19PCh. 21 - Prob. 20PCh. 21 - Prob. 21PCh. 21 - 21.22 Write a mechanism that can account for the...Ch. 21 - Prob. 23PCh. 21 - Prob. 24PCh. 21 - Prob. 25PCh. 21 - 21.26 In 1985, T. Katz (Columbia University)...Ch. 21 - When the following molecule was exposed to the...Ch. 21 - During the course of the following Stille...Ch. 21 - 1. In “The Chemistry of... Complex Cross...Ch. 21 - Prob. 2LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
Chlorine has two isotopes, 35Cl and 37Cl; 75.77% of chlorine is 35Cl, and 24.23% is 37Cl. The atomic mass of 35...
Organic Chemistry
44. Calculate the ratio of CH3NH2 to CH3NH3Cl concentration required to create a buffer with pH = 10.24.
Chemistry: A Molecular Approach (4th Edition)
1.6 Read the labels on products used to wash your dishes. What are the names of some chemicals contained in tho...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Which of the following are not acids? CH3COOH CO2 HNO2 HCOOH CCl4
Essential Organic Chemistry (3rd Edition)
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the name of the mechanism where camphor is reduced to isoborneol? Is it Nucleophillic/Electrophilic Addition/Substitution? Give explanation also.arrow_forwardCompound A has the molecular formula C14H25Br and was obtained by thereaction of sodium acetylide with 1,12-dibromododecane. On treatment ofcompound A with sodium amide, compound B (C14H24) was obtained. Ozonolysisof compound B gave the diacid HO2C(CH2)12CO2H. Catalytic hydrogenation ofcompound B over Lindlar palladium gave compound C (C14H26), andhydrogenation over platinum gave compound D (C14H28). Further, C yieldedO=CH(CH2)12CH=O on ozonolysis. Assign structures to compounds A through Dso as to be consistent with the observed transformations.arrow_forwardChemistryarrow_forward
- Toluene contains a benzene ring with three double bonds in it but still does not show any sign of undergoing a reaction with bromine solution. why?arrow_forwardIdentify the class of polymer, based on functional groups, for the polymers in Problems 26.43 and 26.48.arrow_forwardGive reasons :(i) Electrophilic substitution in aromatic amines takes place more readily than benzene.(ii) CH3CONH2 is weaker base than CH3CH2NH2.arrow_forward
- (b) Compound AA has a molecular formula of CsH&O and gives a positive result using Tollen's reagent. The reaction of compound AA with hot acidified potassium permanganate, KMNO4 gives compound BB. The catalytic hydrogenation of compound AA with nickel, Ni produced compound Cc. The reaction of compound BB with ethanamine, CH:CH2NH2 produces compound DD. Sebatian AA mempunyai formula molekul C3He0 dan memberikan keputusan yang positif menggunakan reagen Tollen. Tindak balas sebatian AA dengan larutan panas kalium permanganat, KMNO4 berasid menghasilkan sebatian BB. Penghidrogenan bermangkin bagi sebatian AA dengan nikel, Ni menghasilkan sebatian CC. Tindak balas sebatian BB dengan etanamina, CH3;CH2NH2 menghasilkan sebatian DD. (1) Draw the structural formula of compounds AA, BB, CC and DD. Lukiskan formula struktur bagi sebatian AA, BB, CC dan DD. (i) Name the type of chemical reaction for the formation of compound CC. Namakan jenis tindak balas kimia bagi penghasilan sebatian Cc.arrow_forward22:43 10. Provide the following: the resonance structure of the intermediate that can resonate into the other intermediate that is given the product that results from the given resonance structure Specify which product is the kinetic product and which is the thermodynamic product . OH 50% H₂SO4 H~arrow_forwardCompound P (C2H4) which is an alkene undergoes reaction with HCl to produce compound Q (C2H5Cl). Reaction of compound Q with benzene in the presence of AlCl3 as catalyst produces compound R. Then, nitration of compound R in the presence ofnH2SO4 produces two compounds, S and T. But when compound R is reacted with a hot acidified solution of alkaline KMnO4 gives compound U. Deduce the structure of compounds P to U.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY