
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21.6, Problem 21.14P
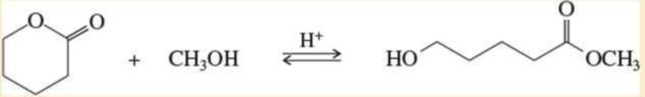
Propose a mechanism for the following ring-opening transesterification. Use the mechanism in Problem21-13 as a model
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
20-23 Predict the product(s) and provide the mechanism for each reaction
below.
NaOH
(a)
CEN
?
H20
(b) N=C-
NaOH
?
H20
(c)
NaOH
CH3-
?
H20
(d)
NaOH
?
N=C-
H20
Ch19-5: Which of the compounds is the intermediate called hemiacetal of the
following reaction?
요
HO OH H₂SO
(-H₂O)
OH
HO
OH
HO
HOO
O
OH
OH
what are the major products off these reactions.
Chapter 21 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 21.2F - Name the following carboxylic acid derivatives,...Ch. 21.4A - Prob. 21.2PCh. 21.4A - Prob. 21.3PCh. 21.4A - Prob. 21.4PCh. 21.5C - Prob. 21.7PCh. 21.6 - When ethyl 4-hydroxybutyrate is heated in the...Ch. 21.6 - Propose a mechanism for the following ring-opening...Ch. 21.6 - Prob. 21.15PCh. 21.7B - Prob. 21.16PCh. 21.7C - Prob. 21.19P
Ch. 21.7C - Prob. 21.20PCh. 21.7C - Prob. 21.21PCh. 21.7D - Prob. 21.22PCh. 21.7D - The mechanism for acidic hydrolysis of a nitrile...Ch. 21.8A - Prob. 21.24PCh. 21.8C - Prob. 21.25PCh. 21.9 - Prob. 21.26PCh. 21.9 - Prob. 21.27PCh. 21.9 - Prob. 21.28PCh. 21.10 - Draw a mechanism for the acylation of anisole by...Ch. 21.10 - Prob. 21.30PCh. 21.11 - Prob. 21.31PCh. 21.11 - Prob. 21.32PCh. 21.12 - Problem 21-33 Propose a mechanism for the...Ch. 21.12 - Suggest the most appropriate reagent for each...Ch. 21.12 - Show how you would synthesize each compound,...Ch. 21.13 - Prob. 21.36PCh. 21.13 - Prob. 21.37PCh. 21.14 - Prob. 21.38PCh. 21.14 - Prob. 21.39PCh. 21.16 - Prob. 21.40PCh. 21.16 - Prob. 21.41PCh. 21 - Prob. 21.42SPCh. 21 - Give appropriate names for the following...Ch. 21 - Predict the major products formed when benzoyl...Ch. 21 - Predict the products of the following reactions....Ch. 21 - Prob. 21.46SPCh. 21 - Prob. 21.47SPCh. 21 - Prob. 21.48SPCh. 21 - Propose mechanisms for the following reactions.Ch. 21 - Prob. 21.51SPCh. 21 - An ether extraction of nutmeg gives large...Ch. 21 - Prob. 21.53SPCh. 21 - Show how you would accomplish the following...Ch. 21 - Prob. 21.55SPCh. 21 - Prob. 21.56SPCh. 21 - Prob. 21.57SPCh. 21 - Prob. 21.58SPCh. 21 - Prob. 21.59SPCh. 21 - Explain this curious result. What does this...Ch. 21 - Prob. 21.61SPCh. 21 - Prob. 21.62SPCh. 21 - Prob. 21.63SPCh. 21 - A chemist was called to an abandoned aspirin...Ch. 21 - Prob. 21.67SPCh. 21 - The IR spectrum, 13ONTVTR spectrum, and 1HNMR...Ch. 21 - Prob. 21.69SPCh. 21 - Prob. 21.70SPCh. 21 - Prob. 21.71SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Naturally occurring compounds called terpenoids, which we'll discuss in Section 27-5, are biosynthesized by a pathway that involves loss of CO2 from 3-phosphomevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this reaction.arrow_forwardReaction of the given D-aldose below with Ac₂0 can be catalyzed by BF3-Et20. Which of the following is the B-furanose structure of the product upon acetylation of the given aldose? Select one: CH₂OAc Aco OAc CH₂OAc OAC AC OAc O CH₂OAC OAc ỌAc ACO OAC OAc O CH,OAc ỌAC OAc HO CHO -OH -ОН CH₂OHarrow_forwardShow how you could synthesize racemic ibuprofen starting with 1-bromo-4-isobutylbenzene.arrow_forward
- Please help with part D attached:arrow_forwardA(n) A(n) H₂N- H | O || H₂N -CICIO _ HO ||| H Glycine OH OH OH T CH₂-C-CH₂ H Glycerol H O H H CH₂ Glycylalanine C-N-C + H₂N- C-O HO T || CICIO CH₂ Alanine reaction converts glycylalanine to glycine and alanine. This reaction requires OH O OH I I CH₂-C-CH₂ Dihydroxyacetone reaction converts glycerol to dihydroxyacetone. This reaction requires and also producesarrow_forwardСНЗСН2СH2CH2CH2CH2CH2ОH plus Н2SO4 produces Нехene Нeptene О Нехупе Нeptynearrow_forward
- SHOW/IDENTIFY the arrow pushing mechanism in at least 4 steps. CH,OPO?- | 3* C=0 2 HO-CH2 1 CH,OPO | 1 C=0 2 Dihydroxyacetone phosphate (DHAP) Но—С H 3 aldolase + Н—С-—ОН H Н-С-—ОН 15 CH-ОРО 6. Н—С—ОН CH,OPO Fructose- 1,6-bisphosphate (FBP) Glyceraldehyde- 3-phosphate (GAP)arrow_forwardIf the following compound is saponified with sodium hydroxide, the products are: O || CH3(CH2) 14CH2-C-O-CH₂CH3 an ester and an alcohol an alcohol and a salt an acid and a salt an acid and an alcohol The purpose of the acid catalyst in the hydrolysis of an amide is: to enhance the nucleophilicity of the water molecule to enhance the electrophilicity of the amide carbonyl carbon to enhance the electrophilicity of the water molecule to shift the equilibrium of the reaction Which of the following compounds has the highest boiling point? CH3CH3 CH3CH2OH CH3-0-CH3 CH3COOHarrow_forwardHow to carry out the following transformation? OCH3 CO2CH3 Co214arrow_forward
- For the substituted cyclohexane compound shown, identify the atoms that will sterically interact with the methyl group in a 1,3‑diaxial fashion.arrow_forwardPenicillins and other β-lactam antibiotics typically develop a resistance to bacteria due to bacterial synthesis of β-lactamase enzymes. Tazobactam, however, is able to inhibit the activity of the β-lactamase by trapping it, thereby preventing resistance from developing. Draw curved arrows to show the movement of electrons in this step of the reaction mechanism.arrow_forward(b) Cortisone OH H2C CH -HO- H3C Oalcohol O carbon-carbon double bond Oester Oaldehyde Ocarbon-carbon triple bond Oether O amide Ocarboxylate anion Oketone Oamine O carboxylic acid O thiol (c) Prostaglandin PGE2 COOH Но OH Oalcohol O carbon-carbon double bond Oester Oaldehyde Ocarbon-carbon triple bond Oether Oketone O amide O carboxylate anion Oamine Ocarboxylic acid O thiolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY