
Concept explainers
Interpretation:
The molecular formula and the condensed structural formula for the given organic compounds are to be given.
Concept Introduction:
The molecular formula indicates the total number of atoms involved in a molecule of a substance.
The condensed structural formula of a molecule does not contain any branching above or below. It is a system of writing organic structures in a line of text.
Answer to Problem 1PE
Solution:
a)
b)
c)
Explanation of Solution
a)

The molecular formula of the given organic compound consists of 6 carbon atoms and
The condensed structural formula for the given organic compound contains 6 carbon atoms and
b)
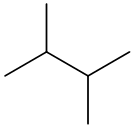
The molecular formula of the given organic compound consists of 6 carbon atoms and
The condensed structural formula for the given organic compound contains 6 carbon atoms and
All carbon and hydrogen atoms are linked together by a single bond and it can be represented as follows:
c)
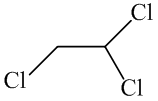
The molecular formula of the given organic compound consists of two carbon atoms,
The condensed structural formula for the given organic compound contains 2 carbon atoms,
Want to see more full solutions like this?
Chapter 22 Solutions
EBK STUDENT SOLUTIONS MANUAL TO ACCOMPA
- Write the condensed structures of all isomers with the formula C2H6O2. Label the functional group (or groups) of each isomer.arrow_forwardName and show FOUR isomers for C3H7NO. Restriction: the molecule can NOT be an alkene, nor can it be an amide. Please state the condensed structures for each isomer. Example of Condesed Structure would look like the image attached.arrow_forwardGive the structural, abbreviated structural and skeletal formulas for the linear hydrocarbon with the molecular formula C4H10. You should either use the 'Building organic molecules' tool in Activity 1.1 of Topic 9 to draw the structural formula of the compound, or take a photograph of a hand-drawn structural formula that you have produced.arrow_forward
- Write the condensed structures of both isomers with the formula C2H6O. Label the functional group of each isomer.arrow_forwardHi, here's my question. Write the condensed structural formulas for two alkenes and one alkyne that all have the molecular formula C6H10.arrow_forwardExplain condensed structures for organic compounds ?arrow_forward
- For the line structure of the alkane shown below write the condensed structural formula.arrow_forwardDraw the condensed structures of the compounds formedfrom (a) butanoic acid and methanol, (b) benzoic acid and2-propanol, (c) propanoic acid and dimethylamine. Namethe compound in each case.arrow_forwardChoose the functional group represented by the condensed structural formula (CH3)2CHCHO.arrow_forward
- Give the molecular formula of a hydrocarbon containingfive carbon atoms that is (a) an alkane, (b) a cycloalkane,(c) an alkene, (d) an alkyne.arrow_forwardQ1: Draw all of the structural isomers possible for the alkane with the molecular formula CeH14, and write the naming of all compounds.arrow_forwardAn organic compound (A) has a general formula CnH2n. This compound has five carbon atoms. When the compound is hydrogenated, it forms a compound (B) having two excess H atoms. a) Identify the compound (A), write its name. b) Identify the compound (B), write its name. c) Which out of (A) and (B) is a saturated hydrocarbon? d) Which of the following figures can be regarded as an Isomer of (B)? ye-cny -CHg-CH-Cly Figure- C43 Fgurea C-2H-cr-cly yc--cns HC igurearrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


