
Organic Chemistry - With Access (Looseleaf) (Custom)
4th Edition
ISBN: 9781259726224
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.23P
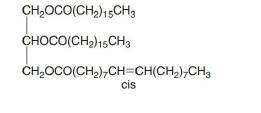
What is the composition of the soap prepared by hydrolysis of the following triacylglycerol?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Organic Chemistry - With Access (Looseleaf) (Custom)
Ch. 22 - Prob. 22.1PCh. 22 - Draw the three possible resonance structures for...Ch. 22 - Prob. 22.3PCh. 22 - Give an IUPAC or common name for each compound.Ch. 22 - Problem 22.5 Draw the structure corresponding to...Ch. 22 - Prob. 22.6PCh. 22 - How would the compounds in each pair differ in...Ch. 22 - Problem 22.8 Deduce the structures of compounds ...Ch. 22 - Prob. 22.9PCh. 22 - Rank the compounds in each group in order of...
Ch. 22 - Prob. 22.11PCh. 22 - Prob. 22.12PCh. 22 - Prob. 22.13PCh. 22 - Prob. 22.14PCh. 22 - Problem 22.15 Draw the products of each...Ch. 22 - Problem 22.16 Draw the products of each reaction.
...Ch. 22 - Prob. 22.17PCh. 22 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.19PCh. 22 - Problem 22.20 Fenofibrate is a...Ch. 22 - Problem 22.21 What product is formed when the...Ch. 22 - How would you synthesize olestra from sucrose?
Ch. 22 - What is the composition of the soap prepared by...Ch. 22 - Problem 22.24 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.25PCh. 22 - Problem 22.26 Some penicillins cannot be...Ch. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Problem 22.30 Glucosamine is a dietry supplement...Ch. 22 - Draw the products of each reaction. a. c. b.Ch. 22 - Draw a tautomer of each compound.Ch. 22 - Draw the product of each reaction. a.b.Ch. 22 - Draw the product of each reaction. a. b.Ch. 22 - Prob. 22.35PCh. 22 - Problem 22.36 Outline two different ways that can...Ch. 22 - 22.37 Rank the following compounds in order of...Ch. 22 - Prob. 22.38PCh. 22 - Prob. 22.39PCh. 22 - Prob. 22.40PCh. 22 - Give thestructure corresponding to each name. a....Ch. 22 - Prob. 22.42PCh. 22 - Prob. 22.43PCh. 22 - 22.43 Explain why is a stronger acid and a weaker...Ch. 22 - Draw the product formed when pentanoyl chloride...Ch. 22 - Draw the product formed when pentanoic anhydride...Ch. 22 - Draw the product formed when phenylacetic acid is...Ch. 22 - Prob. 22.48PCh. 22 - Prob. 22.49PCh. 22 - Draw the product formed when phenylacetonitrile ...Ch. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - Prob. 22.54PCh. 22 - Prob. 22.55PCh. 22 - Prob. 22.56PCh. 22 - Draw a stepwise mechanism for each reaction. a. b.Ch. 22 - When acetic acid CH3COOH is treated with a trace...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - Prob. 22.62PCh. 22 - Prob. 22.63PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - 22.63 Acid-catalyzed hydrolysis of forms compound...Ch. 22 - Prob. 22.66PCh. 22 - Devise a synthesis of each compound using...Ch. 22 - Convert 1-bromohexane (CH3CH2CH2CH2CH2CH2Br) into...Ch. 22 - Prob. 22.69PCh. 22 - Prob. 22.70PCh. 22 - Prob. 22.71PCh. 22 - Prob. 22.72PCh. 22 - Prob. 22.73PCh. 22 - Prob. 22.74PCh. 22 - Devise a synthesis of each labeled compound using...Ch. 22 - 22.70 What polyester or poly amide can be prepared...Ch. 22 - 22.71 What two monomers are needed to prepare each...Ch. 22 - Prob. 22.78PCh. 22 - How can IR spectroscopy be used to distinguish...Ch. 22 - Rank the compounds in each group in order of...Ch. 22 - 22.75 Identify the structures of each compound...Ch. 22 - 22.76 Identify the structures of A and B, isomers...Ch. 22 - Prob. 22.83PCh. 22 - 22.78 Identify the structure of compound C...Ch. 22 - 22.79 Identify the structures of D and E, isomers...Ch. 22 - 22.80 With reference to amides A and B, the...Ch. 22 - Prob. 22.87PCh. 22 - Prob. 22.88PCh. 22 - Prob. 22.89PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - Draw a stepwise mechanism for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What are the four structural subunits that contribute to the structure of a triacylglycerol?arrow_forwardDraw block diagram structures for the three different triacylglycerols that can be produced from glycerol, palmitic acid, stearic acid, and linolenic acid.arrow_forwardDraw block diagram structures for the four different triacylglycerols that can be produced from glycerol, stearic acid, and linolenic acid.arrow_forward
- What additional products are formed when a triacyl-glyoerol molecule is converted to each of the following in an acidic hydrolysis reaction. a. diacylglycerol b. monoacylglycerol c. glycerolarrow_forwardDraw condensed structural formulas for all products you would obtain from the saponification with KOH of the triacylglycerol in Problem 19-54.arrow_forwardWhy can only unsaturated triacylglycerols undergo hydrogenation?arrow_forward
- What are the products of the complete hydrolysis of a triacylglycerol?arrow_forwardWhich of the terms triacylglycerol, glycerophospholipid, and sphingophospholipid applies to each of the following characterizations? More than one term may apply, or none of the terms may apply in a given situation. a. Must contain choline b. Lecithin c. Must contain an ester linkage d. Saponifiablearrow_forwardDraw the structure of a triglyceride that contains one myristic acid, one palmitoleic acid, and one linoleic acid. Identify the ester bonds.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY