
(a)
Interpretation: The reagents required to convert phenylacetonitrile
Concept introduction: Grignard reagents are
Answer to Problem 22.35P
The reagents required to convert phenylacetonitrile
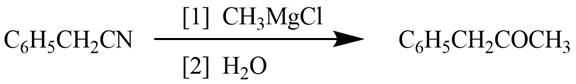
Explanation of Solution
Grignard reagents converts nitriles to corresponding carbonyl groups.
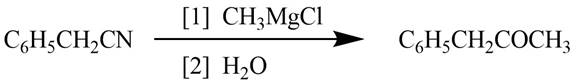
Figure 1
The reagents required to convert phenylacetonitrile
(b)
Interpretation: The reagents required to convert phenylacetonitrile
Concept introduction: Grignard reagents are organometallic compounds having the general formula
Answer to Problem 22.35P
The reagents required to convert phenylacetonitrile
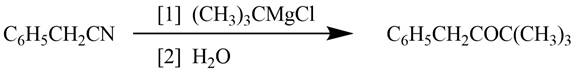
Explanation of Solution
Grignard reagents converts nitriles to corresponding carbonyl groups.
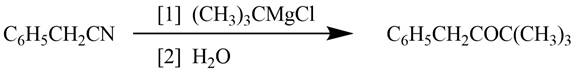
Figure 2
The reagents required to convert phenylacetonitrile
(c)
Interpretation: The reagents required to convert phenylacetonitrile
Concept introduction: Nitrile groups get converted to carbonyl group in presence of reducing agents like DIBAL-H.
Answer to Problem 22.35P
The reagents required to convert phenylacetonitrile
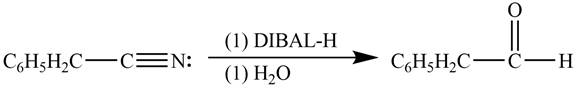
Explanation of Solution
Diisobutylaluminiumhydride(DIBAL-H) is a selective reducing agent. It readily converts nitriles to carbonyl group. DIBAL-H can be used to convert phenylacetonitrile
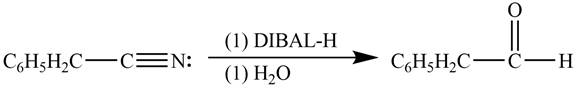
Figure 3
The reagents required to convert phenylacetonitrile
(d)
Interpretation: The reagents required to convert phenylacetonitrile
Concept introduction: Hydrolysis of nitriles in acidic medium convert them to the corresponding carboxylic acids.
Answer to Problem 22.35P
The reagents required to convert phenylacetonitrile
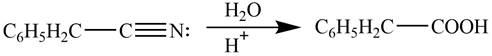
Explanation of Solution
Acidic hydrolysis of phenylacetonitrile
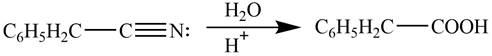
Figure 4
The reagents required to convert phenylacetonitrile
Want to see more full solutions like this?
Chapter 22 Solutions
ORGANIC CHEMISTRY(LL)W/ACCESS>CUSTOM<
- Eleostearic acid, C18H30O2, is a rare fatty acid found in the tung oil used for finishing furniture. On ozonolysis followed by treatment with zinc, eleostearic acid furnishes one part pentanal, two parts glyoxal (OHC-CHO), and one part 9-oxononanoic acid [OHC(CH2)7CO2H]. What is the structure of eleostearic acid?arrow_forwardThe structure of 4 isomers of an aldotetrose carbohydrate are given. 1) select every structure that is a diastereomer of structure D A, B, or C? 2) select every structure that is a enantiomer of structure C D, B, or A? 3) select every structure that is a stereoisomer of structure D A, B, or Carrow_forward(a) Draw a skeletal structure of the anabolic steroid methenolone from the following description. Methenolone contains the tetracyclic steroid skeleton with a carbonyl group at C3, a hydroxyl at C17, a double bond between C1 and C2, and methyl groups bonded to C1, C10, and C13. (b) Add wedges and dashed wedges for all stereogenic centers with thefollowing information: the configuration at C10 is R, the configuration at C13 is S, the configuration at C17 is S, and all substituents at ring fusions are trans to each other. (c) Draw the structure of Primobolan, the product formed when methenolone is treated with CH3(CH2)5COCl and pyridine. Primobolan is an anabolic steroid that can be taken orally or by injection and has been used illegally by well-known Major League Baseball players.arrow_forward
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardDraw the structure of cholic acid, showing the rings in their chairconformations, and label each methyl group and hydroxy group asaxial or equatorial.arrow_forwardIn a 0.28 mM aqueous solution of trimethylacetic acid (C4H9CO2H), what is the percentage of trimethylacetic acid that is dissociated?arrow_forward
- ) How many chiral centers are present in desosamine? How many stereoisomers are possible for it? How many pairs of enantiomers are possible for it?arrow_forwardCH3-CH2-OH reacts with H+/140C = ? + H2Oarrow_forwardWhat happens when (i) Chlorobenzene is treated with Cl2/FeCl3 (ii) ethyl chloride is treated with AgN02, (iii) 2-bromopentane is treated with alcoholic KOH? Write the chemical equations in support of your answer.arrow_forward
- 3-Methyl-2-hexenoic acid (mixture of E and Z isomers) has been identified as the substance responsible for the odor of human sweat. Synthesize the compound from raw materials that have five carbons or less.arrow_forwardDraw and label the chemical structure of the following compounds and label its replacable Hydrogen atoms 1. KHP 2. Ascorbic acidarrow_forward(a) Write the structures of the following compounds and mark them as chiral or achiral. 4 (i) 2-Bromopentane (ii) 3-Bromopentane (iii) 1-Bromo-2-methylbutane (iv) 2-Chloro-3-methylbutane (b) Identify the asymmetric carbon in the chiral compounds. (c) Write the structure of the other enantiomer of the chiral compounds.arrow_forward

 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



