
Concept explainers
Give the structure corresponding to each name.
a. cyclohexyl propanoate
b. cyclohexanecarboxamide
c.
d. Vinyl acetate
e. Benzoic propanoic anhydride
f.
g. octyl butanoate
h. N, N-dibenzylformamide
(a)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
The structure corresponding to the given name is
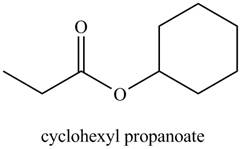
Explanation of Solution
The given name indicates that compound contains ester (name ends with –ate) as the functional group, and propane as the longest carbon chain with one cyclohexyl substitution as
Thus, the structure corresponding to the given name is,
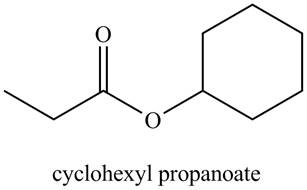
Figure 1
The structure corresponding to the given name is drawn in Figure 1.
(b)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
The structure corresponding to the given name is,
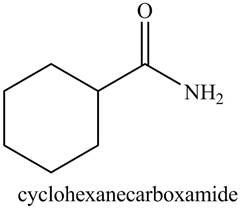
Explanation of Solution
The given name indicates that compound contains amide (name ends with -amide) as the functional group. This amide group is derived from cyclohexylcarboxylic acid, and bonded to cyclohexane ring.
Thus, the structure corresponding to given name is,
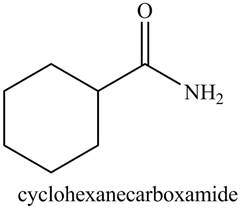
Figure 2
The structure corresponding to the given name is drawn in Figure 2.
(c)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
The structure corresponding to the given name is,
Explanation of Solution
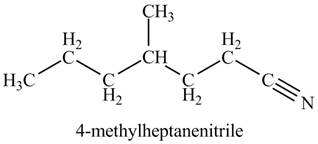
The given name indicates that compound contains cyano (name ends with -nitrile) as the functional group, and heptane as the longest carbon chain with one methyl substitution at
Thus, the structure corresponding to given name is,
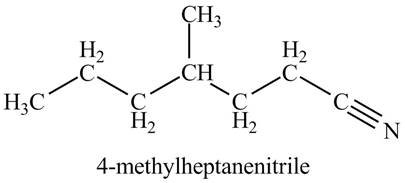
Figure 3
The structure corresponding to the given name is drawn in Figure 3.
(d)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
The structure corresponding to the given name is,
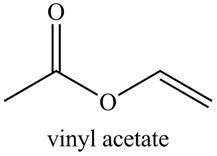
Explanation of Solution
The given name indicates that compound contains ester (name ends with -ate) as the functional group, and an acetate group is bonded to a vinyl group
Thus, the structure corresponding to the given name is,
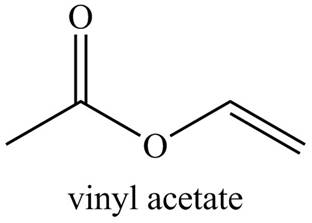
Figure 4
The structure corresponding to the given name is drawn in Figure 4.
Interpretation: The structure corresponding to the given name is to be drawn.
(e)
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
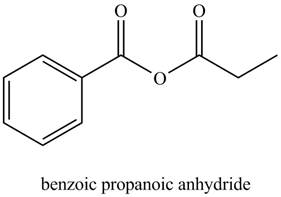
The structure corresponding to the given name is,
Explanation of Solution
The given name indicates that compound contains anhydride (name ends with -anhydride) as the functional group. This anhydride compound is derived from benzoic acid and propanoic acid.
Thus, the structure corresponding to the given name is,

Figure 5
The structure corresponding to the given name is drawn in Figure 5.
Interpretation: The structure corresponding to the given name is to be drawn.
(f)
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
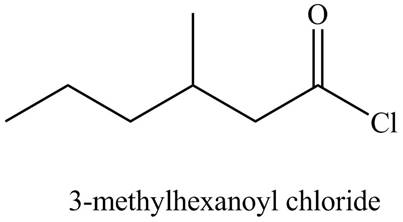
The structure corresponding to the given name is,
Explanation of Solution
The given name indicates that compound contains acid chloride (name ends with -yl chloride) as the functional group, and hexane as the longest carbon chain with one methyl substitution at
Thus, the structure corresponding to the given name is,
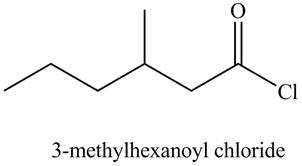
Figure 6
The structure corresponding to the given name is drawn in Figure 6.
Interpretation: The structure corresponding to the given name is to be drawn.
(g)
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
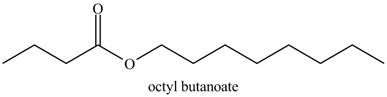
The structure corresponding to the given name is,
Explanation of Solution
The given name indicates that compound contains ester (name ends with –ate) as the functional group, and butane as the longest carbon chain with one octyl substitution as
Thus, the structure corresponding to the given name is,

Figure 7
The structure corresponding to the given name is drawn in Figure 7.
(h)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.41P
The structure corresponding to the given name is,
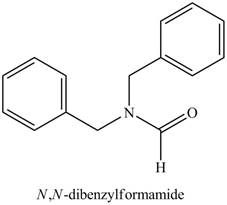
Explanation of Solution
The given name indicates that compound contains amide (name ends with -amide) as the functional group. This amide group is derived from formic acid, and bonded to two benzyl groups.
Thus, the structure corresponding to the given name is,
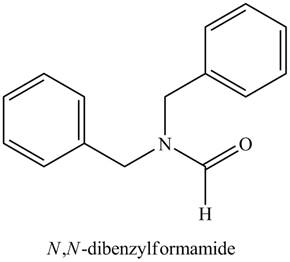
Figure 8
The structure corresponding to the given name is drawn in Figure 8.
Want to see more full solutions like this?
Chapter 22 Solutions
ORGANIC CHEMISTRY LL W/SSM+CONNECT+KIT
- Which of the following will not oxidize benzaldehyde? -- acidified KMnO4 -- acidified K2CrO4 -- Tollen’s reagent followed by H3O+ -- Basic I2 in KIarrow_forwardGive the necessary reagents for each part in the correct order.arrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Why does the Ritter reaction not work with primary alcohols? b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.arrow_forward
- What methods introduce the functional groups in the product ?arrow_forwardWhy some substituents make a benzene ring react faster than benzeneitself (activators) ?arrow_forwardis ethyl acetate and benzoic acid in NaHCO3 soluble or insoluble? Please explain why lIs cyclohexene and etyhylamine soluble/insoluble in NaOH? Please explain why. Is Acetic anhydride soluble/ insoluble in HCl? Why? Is H2SO4 soluble/insoluble in Cyclohexene? Please explain whyarrow_forward
- 1. Give the major product for the reactions in d and e. 2. Give the name of the structure in f.arrow_forwardneed help with providing a good reagentarrow_forwarda) Put these three common types of carbonyl compound in order of decreasing reactivity ester amide acid chloride b) For the least reactive, show the interconversion to its other resonance form: How does this electron delocalisation make it stable? c) For the most reactive, draw the mechanism of its undergoing hydrolysis (reaction with H2O): Why makes this type of carbonyl so reactive to nucleophiles?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





