
Concept explainers
Draw the product formed when phenylacetonitrile
a.
b.
c.
d.
e.
f.
(a)
Interpretation: The product formed from the treatment of phenylacetonitrile
Concept introduction: Hydrolysis of nitriles in acidic medium converts them to the corresponding carboxylic acids. The reaction involves two parts; that is nucleophilic addition followed by nucleophilic acyl substitution.
Answer to Problem 22.46P
The product formed from the treatment of phenylacetonitrile
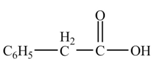
Explanation of Solution
The given compound is a nitrile.
Hydrolysis of nitriles in acidic medium converts them to the corresponding carboxylic acids. The reaction involves two parts; that is nucleophilic addition followed by nucleophilic acyl substitution. Phenylacetonitrile
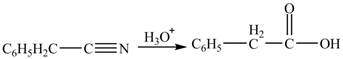
Figure 1
The product formed from the treatment of phenylacetonitrile
(b)
Interpretation: The product formed from the treatment of phenylacetonitrile
Concept introduction: Phenylacetonitrile undergo hydrolysis in basic medium to form corresponding carboxylate ion. The mechanism of the reaction involves two parts. The first part is conversion of nitrile to a
Answer to Problem 22.46P
The product formed from the treatment of phenylacetonitrile
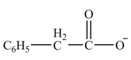
Explanation of Solution
The given compound is a nitrile.
Hydrolysis of nitriles in basic medium converts them to the corresponding carboxylate anion. Nitriles react with
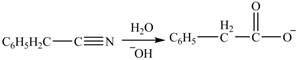
Figure 2
The product formed from the treatment of phenylacetonitrile
(c)
Interpretation: The product formed from the treatment of phenylacetonitrile
Concept introduction: The Grignard reagents are organometallic compounds having the general formula
Answer to Problem 22.46P
The product formed from the treatment of phenylacetonitrile
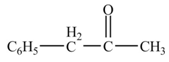
Explanation of Solution
The given compound is a nitrile.
Nitriles react with both Grignard reagent, and organolithium reagents, followed by hydrolysis to yield ketones. The reaction utilizes
Phenylacetonitrile undergoes Grignard reaction with
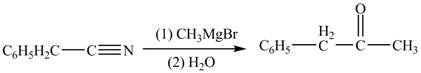
Figure 3
The product formed from the treatment of phenylacetonitrile
(d)
Interpretation: The product formed from the treatment of phenylacetonitrile
Concept introduction: Organolithium reagents are organometallic compounds having the general formula
Answer to Problem 22.46P
The product formed from the treatment of phenylacetonitrile
Explanation of Solution
The given compound is a nitrile.
Nitriles react with both Grignard reagent, and organolithium reagents, followed by hydrolysis to yield ketones. The reaction utilizes
The treatment of phenylacetonitrile with
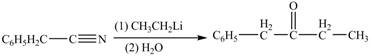
Figure 4
The product formed from the treatment of phenylacetonitrile
Interpretation: The product formed from the treatment of phenylacetonitrile
(e)
Concept introduction:
Answer to Problem 22.46P
The product formed from the treatment of phenylacetonitrile
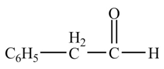
Explanation of Solution
Diisobutylaluminiumhydride
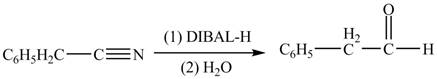
Figure 5
The product formed from the treatment of phenylacetonitrile
(f)
Interpretation: The product formed from the treatment of phenylacetonitrile
Concept introduction:
Answer to Problem 22.46P
The product formed from the treatment of phenylacetonitrile
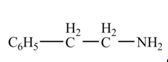
Explanation of Solution
Nitriles react with
![]()
Figure 6
The product formed from the treatment of phenylacetonitrile
Want to see more full solutions like this?
Chapter 22 Solutions
ORG.CHEMISTRY CONNECT ACCESS>CUSTOM<
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. a. HNO3, H2SO4 h. product in (a), then Sn, HClarrow_forwardDraw the products formed when D-altrose is treated with each reagent. a. (CH3)2CHOH, HCl b. NaBH4, CH3OH c. Br2, H2O d. HNO3, H2O e. [1] NH2OH; [2] (CH3CO)2O, NaOCOCH3; [3] NaOCH3 f. [1] NaCN, HCl; [2] H2, Pd-BaSO4; [3] H3O+ g. CH3I, Ag2O h. C6H5CH2NH2, mild H+arrow_forward
- Draw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] HC≡CNa; [2] H2Oarrow_forwardThioglycolic acid, HSCH2CO2H, a substance used in depilatory agents (hair removers) has pKa = 3.42. What is the percent dissociation of thioglycolic acid in a buffer solution at pH = 3.0?arrow_forwardWhat product is formed when CH3OCH2CH2C ≡ CCH2CH(CH3)2 is treated with each reagent: (a) H2 (excess), Pd-C; (b) H2(1 equiv), Lindlar catalyst; (c) H2 (excess), Lindlar catalyst; (d) Na, NH3?arrow_forward
- Draw the products when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents no reaction occurs: A)NaCl B)NH3(1 eqiv) C)1)CH2NH2, 2)CH3COClarrow_forwardDraw the product formed when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents, no reaction occurs. a. NaHCO3 b. NaOH c. SOCl2 d. NaCl e. NH3(1equiv) f. NH3, ∆ g. CH3OH, H2SO4 h. CH3OH, −OH i. [1] NaOH; [2] CH3COCl j. CH3NH2, DCC k. [1] SOCl2; [2] CH3CH2CH2NH2 (excess) l. [1] SOCl2; [2] (CH3)2; [2] (CHarrow_forward#20 B Draw structural formulas for all possible carbocations formed by the reaction of each alkene with HCl.arrow_forward
- Draw the products formed when (CH3)2C=CH2 is treated with following reagent. [1] BH3; [2] H2O2, HO−arrow_forwardDraw the organic products formed when allylic alcohol A is treated with each reagent.a.H2 + Pd-C b.mCPBA c. PCC d.CrO3, H2SO4, H2O e.(CH3)3COOH, Ti[OCH(CH3)2]4, (+)-DET f. (CH3)3COOH, Ti[OCH(CH3)2]4, (−)-DET g. [1] PBr3; [2] LiAlH4; [3] H2O h.HCrO4−–Amberlyst A-26 resinarrow_forwardDraw the products formed from nitration of each compoundarrow_forward
