
Concept explainers
(a)
Interpretation: The crystal field splitting of the given structures to be interpreted.
Concept Introduction:
Spectrochemical series: The list of ligands arranged in an ascending order of
Crystal field splitting: The energy gap between the splitting of d-orbitals of the metal ion in presence of ligands is known as the crystal field splitting
To Identify: The crystal field splitting of the given structures to be interpreted.
(a)
Explanation of Solution
Interpret the reason.
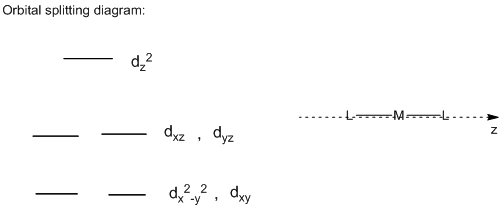
Orbital Splitting diagram of linear complex:
In the linear structure, the ligands which are pointing directly towards the z-axis, the
(b)
Interpretation: The crystal field splitting of the given structures to be interpreted.
Concept Introduction:
Spectrochemical series: The list of ligands arranged in an ascending order of
Crystal field splitting: The energy gap between the splitting of d-orbitals of the metal ion in presence of ligands is known as the crystal field splitting
To Identify: The crystal field splitting of the given structures to be interpreted.
(b)
Explanation of Solution
Interpret the reason.
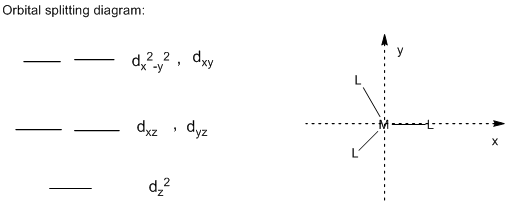
Orbital Splitting diagram of trigonal complex:
In the trigonal structure, the ligands which are pointing directly towards the x-axis and y-axis, the
(c)
Interpretation: The crystal field splitting of the given structures to be interpreted.
Concept Introduction:
Spectrochemical series: The list of ligands arranged in an ascending order of
Crystal field splitting: The energy gap between the splitting of d-orbitals of the metal ion in presence of ligands is known as the crystal field splitting
To Identify: The crystal field splitting of the given structures to be interpreted.
(c)
Explanation of Solution
Interpret the reason.
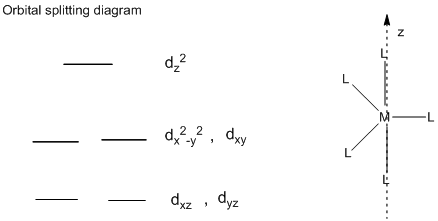
Orbital Splitting diagram of trigonal bipyramidal complex:
In the trigonal bipyramidal structure, the ligands which are pointing directly towards the z-axis, the
Want to see more full solutions like this?
Chapter 22 Solutions
Package: Chemistry - Atoms First With Aleks 360 Access Card (1-semester)
- Four different octahedral chromium coordination compounds exist that all have the same oxidation state for chromium and have H2O and Cl as the ligands and counterions. When 1 mole of each of the four compounds is dissolved in water, how many moles of silver chloride will precipitate upon addition of excess AgNO3?arrow_forwardWhich of the following statement(s) is( are) true? a. The coordination number of a metal ion in an octahedral complex ion is 8. b. All tetrahedral complex ions are low-spin. c. The formula for triaquatrianuninechromiwn(III) sulfate is [Cr(H2O)3(NH3)](SO4)3. d. The electron configuration of Hf2+ is [Xe]4f126s2. e. Hemoglobin contains Fe3+.arrow_forwardHow many geometric isomers are possible for the square-planar complex ion [Pt(NH3)(CN)Cl2)?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





