
Concept explainers
Identify the structures of each compound from the given data.
a. Molecular formula
IR absorption:
(quartet,
b. Molecular formula
IR absorption:
(triplet,
c. Molecular formula
IR absorption:
d. Molecular formula
IR absorption:
(triplet,
e. Molecular formula
IR absorption:
(a)
Interpretation: The structure of compound that has molecular formula
Concept introduction: The method of spectroscopy is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 22.75P
The structure of given compound is,
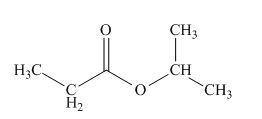
Explanation of Solution
The IR absorption value of the given compound is
A septet of one hydrogen atoms at
A triplet peak of three hydrogen atoms and a quartet of two carbon atoms shows that their carbon atoms are bonded to each other and the splitting peak takes place according to
Therefore, the structure of compound is,

Figure 1
The structure of given compound is shown in Figure 1.
(b)
Interpretation: The structure of compound that has molecular formula
Concept introduction: The method of spectroscopy is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 22.75P
The structure of given compound is,
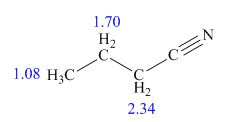
Explanation of Solution
The IR absorption value of the given compound is
In the given compound, one out of four carbon atoms is
Therefore, the structure of compound is,

Figure 2
The structure of given compound is shown in Figure 2.
(c)
Interpretation: The structure of compound that has molecular formula
Concept introduction: The method of spectroscopy is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 22.75P
The structure of given compound is,
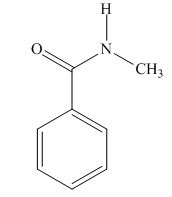
Explanation of Solution
The IR absorption value of the given compound are
Multiple
A singlet NMR peak of one hydrogen atom is observed due to presence of
Therefore, the structure of compound is,

Figure 3
The structure of given compound is shown in Figure 3.
(d)
Interpretation: The structure of compound that has molecular formula
Concept introduction: The method of spectroscopy is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 22.75P
The structure of given compound is,
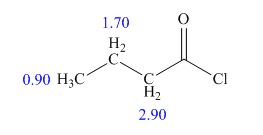
Explanation of Solution
The IR absorption value of the given compound is
In the given compound, one out of four carbon atoms is
Therefore, the structure of compound is,

Figure 4
The structure of given compound is shown in Figure 4.
(e)
Interpretation: The structure of compound that has molecular formula
Concept introduction: The method of spectroscopy is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 22.75P
The structure of given compound is,
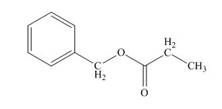
Explanation of Solution
The IR absorption value of the given compound is
Multiple
Therefore, the structure of compound is,
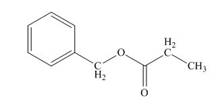
Figure 5
The structure of given compound is shown in Figure 5.
Want to see more full solutions like this?
Chapter 22 Solutions
ORGANIC CHEMISTRY LSLF W/CONNECT >BI<
- Following are 1H-NMR spectra for compounds G, H, and I, each with the molecular formula C5H12O. Each is a liquid at room temperature, is slightly soluble in water, and reacts with sodium metal with the evolution of a gas. (a) Propose structural formulas of compounds G, H, and I. (b) Explain why there are four lines between 0.86 and 0.90 for compound G. (c) Explain why the 2H multiplets at 1.5 and 3.5 for compound H are so complex.arrow_forwardPropose a structural formula for each compound consistent with its 1H-NMR and 13C-NMR spectra. (a) C5H10O2 (b) C7H14O2 (c) C6 H12O2 (d) C7H12O4 (e) C4H7ClO2 (f) C4H6O2arrow_forwardPropose a structure consistent with each set of spectral data: a.C6H14O: IR peak at 3600−3200 cm−1; NMR (ppm): 0.8 (triplet, 6 H) 1.5 (quartet, 4 H) 1.0 (singlet, 3 H) 1.6 (singlet, 1 H) b.C6H14O: IR peak at 3000−2850 cm−1; NMR (ppm): 1.10 (doublet, relative area = 6) 3.60 (septet, relative area = 1)arrow_forward
- Propose a structure consistent with each set of data Compound A: Molecular formula: C8H10OIR absorption at 3150−2850 cm−11H NMR data: 1.4 (triplet, 3 H), 3.95 (quartet, 2 H), and 6.8–7.3(multiplet, 5 H) ppmarrow_forwardPropose a structure consistent with each set of data.a. Compound A:Molecular formula: C8H10OIR absorption at 3150–2850 cm–11H NMR data: 1.4 (triplet, 3 H), 3.95 (quartet, 2 H), and 6.8–7.3 (multiplet, 5 H) ppm b. Compound B:Molecular formula: C9H10O2IR absorption at 1669 cm–11H NMR data: 2.5 (singlet, 3 H), 3.8 (singlet, 3 H), 6.9 (doublet, 2 H), and 7.9 (doublet, 2 H) ppmarrow_forwardPropose a structure consistent with following set of spectral data: C5H10O2: IR peak at 1740 cm−1; NMR (ppm): 1.15 (triplet, 3 H) 2.30 (quartet, 2 H) 1.25 (triplet, 3 H) 4.72 (quartet, 2 H)arrow_forward
- Compound A has molecular formula C7H7X. Its 1H-NMR spectrum shows a singlet at 2.26 ppm and two doublets, one at 6.95 ppm and one at 7.28 ppm. The singlet has an integral of three and the doublets each have an integral of two. Its 13C-NMR shows five signals. The mass spectrum of A shows a peak at m/z = 170 and another peak at m/z = 172; the relative height of the two peaks is 1:1 respectively. - Identify what atom X is, explaining your reasoning - Identify Compound A, explaining your reasoning Compound A is treated with a mixture of nitric and sulfuric acids to generate Compound B. The 1H-NMR spectrum of B shows two singlets, one at 2.52 pm and one at 8.13 ppm. The 13C-NMR spectrum of B shows five signals. The mass spectrum of B shows a peak at m/z = 260 and another peak at m/z = 262; the relative height of the two peaks is 1:1 respectively. - Identify compound B, explaining your reasoning Compound B is treated with sodium ethoxide to generate compound C. The 1H-NMR spectrum of C shows…arrow_forwardIdentify below compound from its spectral data. Molecular formula: C4H7N IR:2250 cm−1 1H NMR data:1.08 (triplet, 3 H), 1.70 (multiplet, 2 H), and 2.34 (triplet,2 H) ppmarrow_forwardA hydrocarbon, compound B, has molecular formula C6H6, and gave an NMR spectrum with two signals: delta 6.55 pm and delta 3.84 pm with peak ratio of 2:1. When warmed in pyridine for three hr, compound B quantitatively converts to benzene. Mild hydrogenation of B yielded another compound C with mass spectrum of m/z 82. Infrared spectrum showed no double bonds; NMR spectrum showed one broad peak at delta 2.34 ppm. With this information, address the following questions. a) How many rings are in compound C? b) How many rings are probably in B? How many double bonds are in B? c) Can you suggest a structure for compounds B and C? d) In the NMR spectrum of B, the up-field signal was a quintet, and the down field signal was a triplet. How must you account for these splitting patterns?arrow_forward
- Propose possible structures consistent with each set of data. Assume each compound has an sp3 hybridized C – H absorption in its IR spectrum, and that other major IR absorptions above 1500 cm are listed. a. a compound having a molecular ion at 72 and an absorption in its IR spectrum at 1725 cm−1 b. a compound having a molecular ion at 55 and an absorption in its IR spectrum at ~2250 cm−1 c. a compound having a molecular ion of 74 and an absorption in its IR spectrum at 3600–3200 cm−1arrow_forwardPropose a structure consistent with each set of data. a.Compound J: molecular ion at 72; IR peak at 1710 cm−1; 1H NMR data J: (ppm) at 1.0 (triplet, 3 H), 2.1 (singlet, 3 H), and 2.4 (quartet, 2 H)b. Compound K: molecular ion at 88; IR peak at 3600-3200 cm−1; 1H NMR K: data (ppm) at 0.9 (triplet, 3 H), 1.2 (singlet, 6 H), 1.5 (quartet, 2 H), and 1.6 (singlet, 1 H)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
