
Concept explainers
Propose a structural formula for each compound consistent with its 1H-NMR and 13C-NMR spectra.
- (a) C5H10O2
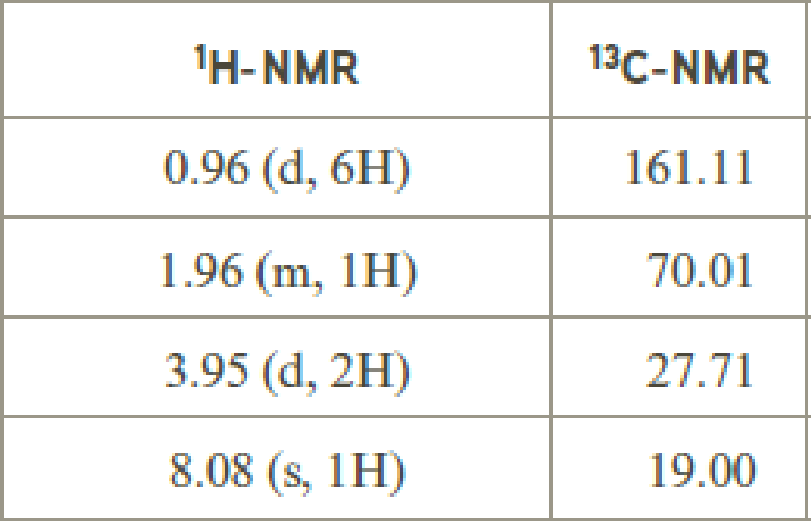
- (b) C7H14O2
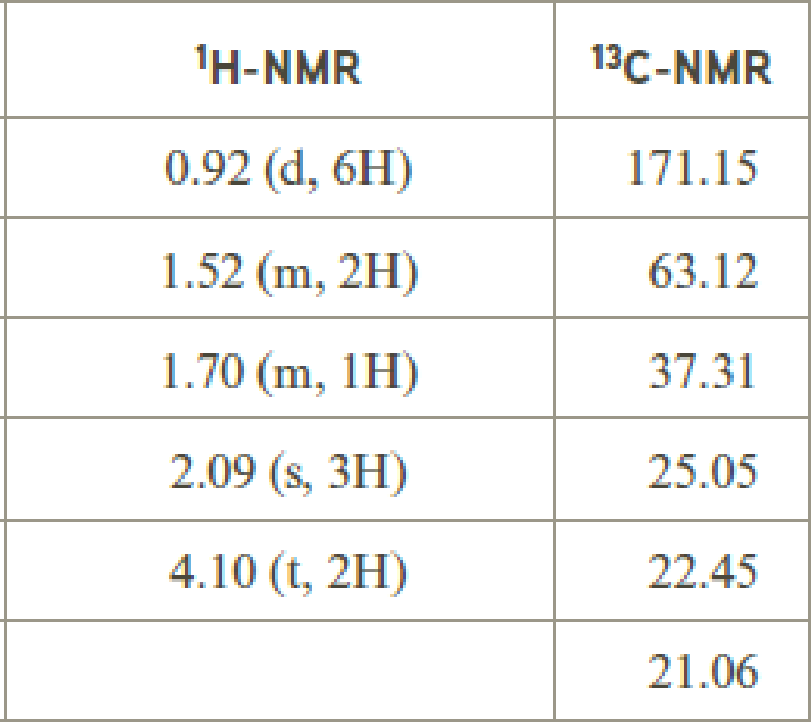
- (c) C6 H12O2
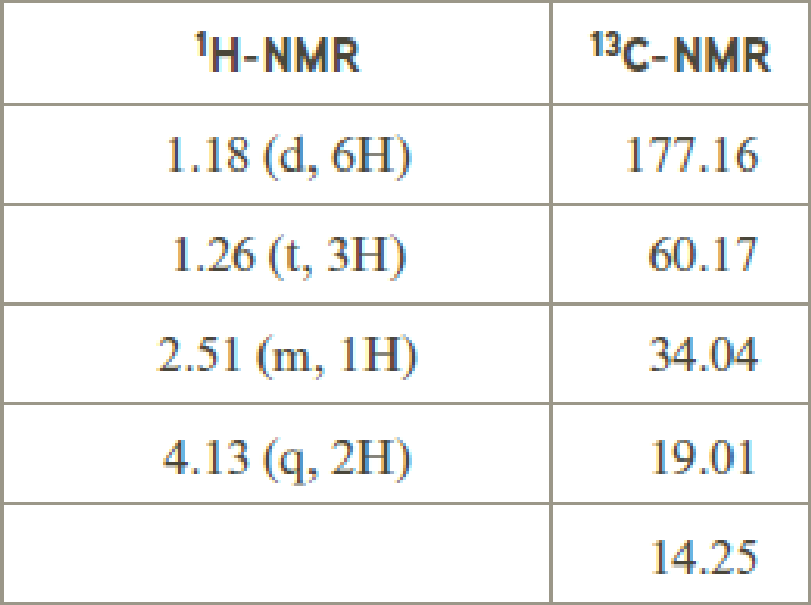
- (d) C7H12O4
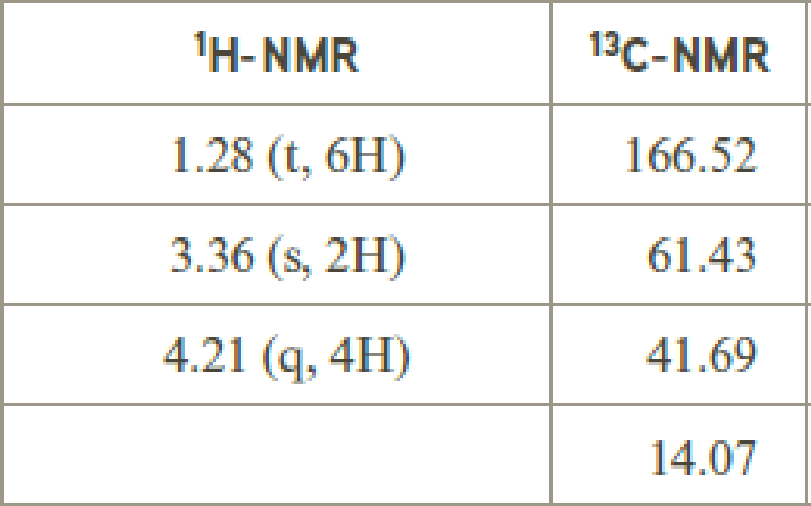
- (e) C4H7ClO2
- (f) C4H6O2
(a)
Interpretation:
The structural formula for compound having molecular formula
Concept Introduction:
It is the study of the interaction between electromagnetic radiation and the nuclei of hydrogen atoms and the hydrogen atoms.
Chemical shift: The frequency of the proton signal in the spectrum with reference to the standard compound which may be TMS(Tetramethylsilane) shows signal at 0 ppm(parts per million).
Integration value (I): The integration value at the bottom of the
Chemical shift values for protons in different electronic environment
| FUNCTIONAL GROUP | EFFECT ON THE ALPHA PROTONS | EFFECT ON THE BETA PROTONS |
| Oxygen of an alcohol or ether | ||
| Oxygen of ester | ||
| Carbonyl groups |
It is the study of the interaction between electromagnetic radiation and the nuclei of the carbon atoms.
The signals in the
Explanation of Solution
Given compound has the molecular formula
Spectral data is given below:
By analyzing the spectral data, the structure can be predicted.
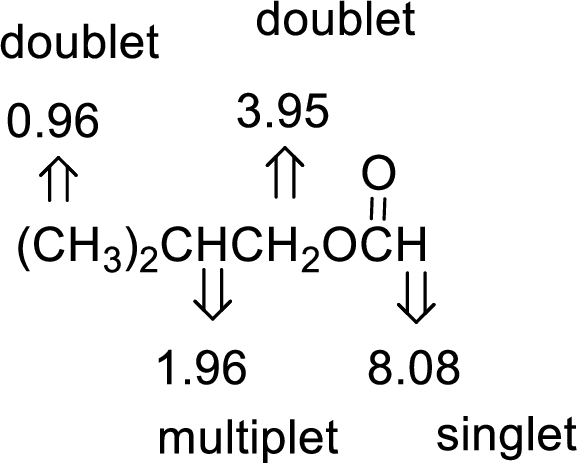
The value of 8.08 suggests an aldehyde proton. 0.96 will indicate the presence of methyl groups and 1.96 indicates the methylene proton
The structure proposed according to the data is given below:
(b)
Interpretation:
The structural formula for compound having molecular formula
Concept Introduction:
It is the study of the interaction between electromagnetic radiation and the nuclei of hydrogen atoms and the hydrogen atoms.
Chemical shift: The frequency of the proton signal in the spectrum with reference to the standard compound which may be TMS(Tetramethylsilane) shows signal at 0 ppm(parts per million).
Integration value (I): The integration value at the bottom of the
Chemical shift values for protons in different electronic environment
| FUNCTIONAL GROUP | EFFECT ON THE ALPHA PROTONS | EFFECT ON THE BETA PROTONS |
| Oxygen of an alcohol or ether | ||
| Oxygen of ester | ||
| Carbonyl groups |
It is the study of the interaction between electromagnetic radiation and the nuclei of the carbon atoms.
The signals in the
Explanation of Solution
Given compound has the molecular formula
Spectral data is given below:
By analyzing the spectral data, the structure can be predicted.
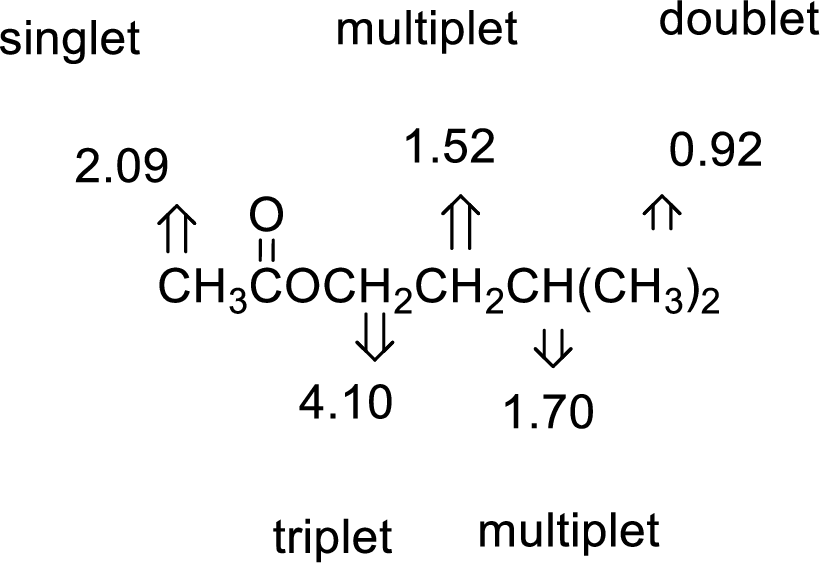
The value of 2.09 indicates a methyl group
The structure proposed according to the data is given below:
(c)
Interpretation:
The structural formula for compound having molecular formula
Concept Introduction:
It is the study of the interaction between electromagnetic radiation and the nuclei of hydrogen atoms and the hydrogen atoms.
Chemical shift: The frequency of the proton signal in the spectrum with reference to the standard compound which may be TMS(Tetramethylsilane) shows signal at 0 ppm(parts per million).
Integration value (I): The integration value at the bottom of the
Chemical shift values for protons in different electronic environment
| FUNCTIONAL GROUP | EFFECT ON THE ALPHA PROTONS | EFFECT ON THE BETA PROTONS |
| Oxygen of an alcohol or ether | ||
| Oxygen of ester | ||
| Carbonyl groups |
It is the study of the interaction between electromagnetic radiation and the nuclei of the carbon atoms.
The signals in the
Explanation of Solution
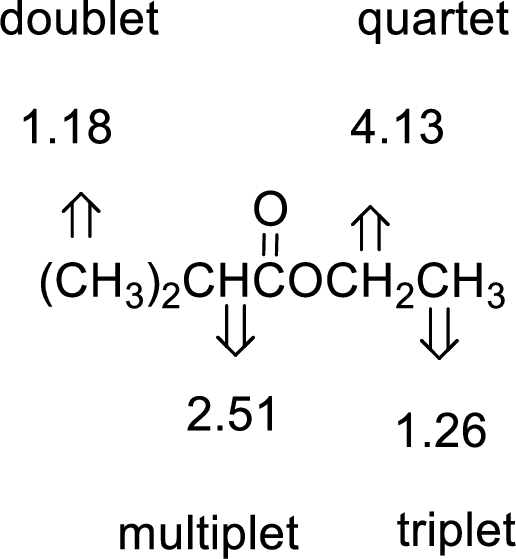
Given compound has the molecular formula
Spectral data is given below:
By analyzing the spectral data, the structure can be predicted.
1.18 and 1.26 will indicate the presence of methyl groups and 4.13 indicates the methylene proton
The structure proposed according to the data is given below:
(d)
Interpretation:
The structural formula for compound having molecular formula
Concept Introduction:
It is the study of the interaction between electromagnetic radiation and the nuclei of hydrogen atoms and the hydrogen atoms.
Chemical shift: The frequency of the proton signal in the spectrum with reference to the standard compound which may be TMS(Tetramethylsilane) shows signal at 0 ppm(parts per million).
Integration value (I): The integration value at the bottom of the
Chemical shift values for protons in different electronic environment
| FUNCTIONAL GROUP | EFFECT ON THE ALPHA PROTONS | EFFECT ON THE BETA PROTONS |
| Oxygen of an alcohol or ether | ||
| Oxygen of ester | ||
| Carbonyl groups |
It is the study of the interaction between electromagnetic radiation and the nuclei of the carbon atoms.
The signals in the
Explanation of Solution
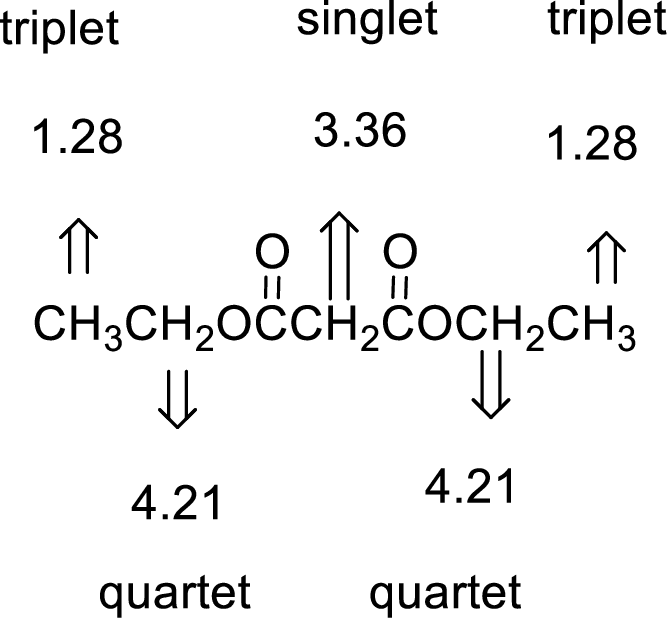
Given compound has the molecular formula
Spectral data is given below:
By analyzing the spectral data, the structure can be predicted.
The value of 3.36 suggests a methylene directly attach to a carbonyl group. 1.28 will indicate the presence of methyl groups and 4.21 indicates the methylene proton
The structure proposed according to the data is given below:
(e)
Interpretation:
The structural formula for compound having molecular formula
Concept Introduction:
It is the study of the interaction between electromagnetic radiation and the nuclei of hydrogen atoms and the hydrogen atoms.
Chemical shift: The frequency of the proton signal in the spectrum with reference to the standard compound which may be TMS(Tetramethylsilane) shows signal at 0 ppm(parts per million).
Integration value (I): The integration value at the bottom of the
Chemical shift values for protons in different electronic environment
| FUNCTIONAL GROUP | EFFECT ON THE ALPHA PROTONS | EFFECT ON THE BETA PROTONS |
| Oxygen of an alcohol or ether | ||
| Oxygen of ester | ||
| Carbonyl groups |
It is the study of the interaction between electromagnetic radiation and the nuclei of the carbon atoms.
The signals in the
Explanation of Solution
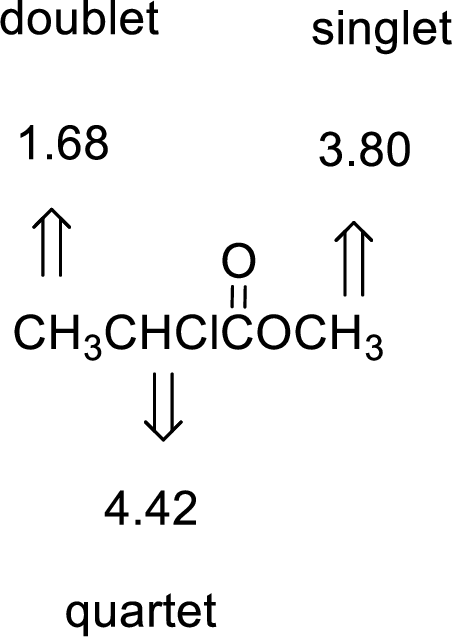
Given compound has the molecular formula
Spectral data is given below:
By analyzing the spectral data, the structure can be predicted.
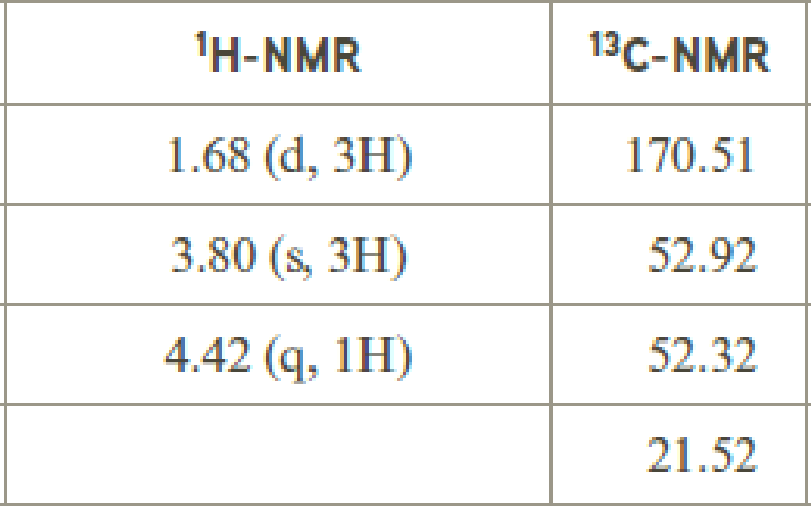
The value of 4.42 suggests a proton group directly attach to a chlorine group. 3.80 will indicate the presence of methyl group which is
The structure proposed according to the data is given below:
(f)
Interpretation:
The structural formula for compound having molecular formula
Concept Introduction:
It is the study of the interaction between electromagnetic radiation and the nuclei of hydrogen atoms and the hydrogen atoms.
Chemical shift: The frequency of the proton signal in the spectrum with reference to the standard compound which may be TMS(Tetramethylsilane) shows signal at 0 ppm(parts per million).
Integration value (I): The integration value at the bottom of the
Chemical shift values for protons in different electronic environment
| FUNCTIONAL GROUP | EFFECT ON THE ALPHA PROTONS | EFFECT ON THE BETA PROTONS |
| Oxygen of an alcohol or ether | ||
| Oxygen of ester | ||
| Carbonyl groups |
It is the study of the interaction between electromagnetic radiation and the nuclei of the carbon atoms.
The signals in the
Explanation of Solution
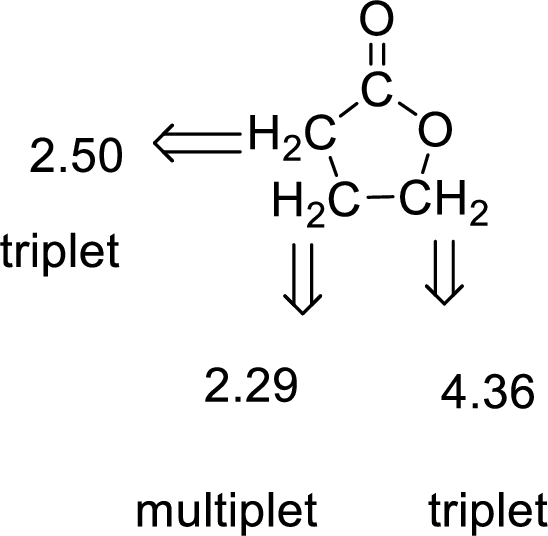
Given compound has the molecular formula
Spectral data is given below:
By analyzing the spectral data, the structure can be predicted.
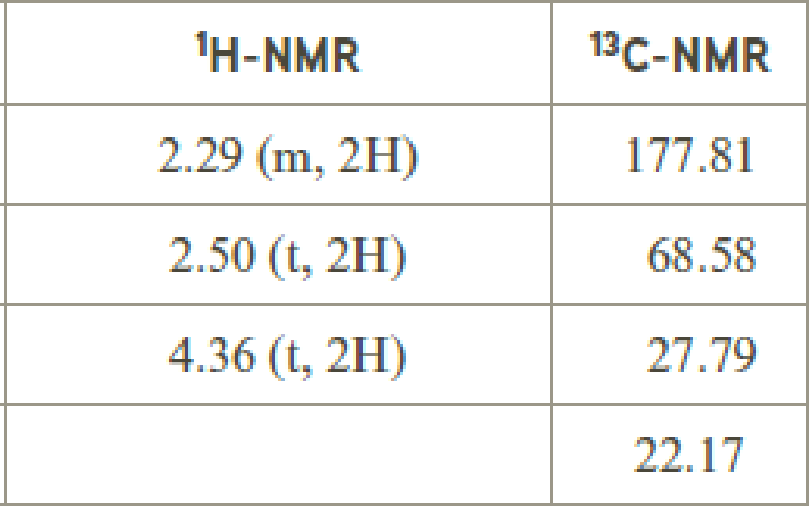
The value of 4.36 suggests a methylene directly attach to a oxygen group. 2.50 will indicate the presence of methylene proton
The structure proposed according to the data is given below:
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Principles of General, Organic, Biological Chemistry
Chemistry (7th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
General, Organic, & Biological Chemistry
- Compound A undergoes an acid-catalyzed hydrolysis. One of the products (B) that is isolated gives the following 1H NMR spectrum. Identify the compounds A and Carrow_forwardPhenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in over-thecounter pain relievers such as APC (aspirin, phenacetin, caffeine), phenacetin is no longer used because of its liver toxicity. Deduce the structure of phenacetin from its 1H NMR and IR spectra.arrow_forwardA compound of formula C11H16N2 gives the IR, 1H NMR, and 13C NMR spectra shown. The proton NMR peak at δ 2 disappears on shaking with D2 Propose a structure for this compound, and show how your structure accounts for the observed absorptions.arrow_forward
- Compound A with molecular formula C6H10 has two peaks in its 1H NMR spectrum, both of which are singlets (with ratio 9 : 1). Compound A reacts with an acidic aqueous solution containing mercuric sulfate to form compound B, which gives a positive iodoform test and has an 1H NMR spectrum that shows two singlets (with ratio 3 : 1). Identify A and B.arrow_forwardPropose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm−1 and the following 1H NMR data:arrow_forwardCompound A has molecular formula C5H8Br4 but shows only one singlet in the 1H-NMR spectrum. Suggest a structure for A and explain your reasoning.arrow_forward
- Reaction of unknown A with HCl forms chlorohydrin B as the major product. A shows no absorptions in its IR spectrum at 1700 cm−1 or 3600-3200 cm−1, and gives the following 1H NMR data: 1.4 (doublet, 3 H), 3.0 (quartet of doublets, 1 H), 3.5 (doublet, 1 H), 3.8 (singlet, 3 H), 6.9 (doublet, 2 H), and 7.2 (doublet, 2 H) ppm. (a) Propose a structure for A, including stereochemistry. (b) Explain why B is the major product in this reaction.arrow_forwardCompound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forwardCompounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forward
- Propose a structure of compound C (molecular formula C10H12O) consistent with the following data. C is partly responsible for the odor and flavor of raspberries. Compound C: IR absorption at 1717 cm-1arrow_forward(a) Compound A has molecular formula C5H10O. It shows three signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.1 ppm, a singlet of integral 3 at 2.14 ppm, and a quintet of integral 1 at 2.58 ppm. Suggest a structure for A and explain your reasoning. (b) Compound B has molecular formula C8H6O2. The IR, 1H-NMR, and 13C-NMR spectra are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Suggest a structure for B and explain your reasoning. (c) Compound C has molecular formula C5H8O. The IR, mass, 1H-NMR, and 13C-NMR spectra are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Suggest a structure for C and explain your reasoning.arrow_forwardPropose a structure for a compound of molecular formula C3H8O with an IR absorption at 3600–3200 cm−1 and the following NMR spectrum:arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
