
Chemistry: Central Science - Package (Custom)
13th Edition
ISBN: 9781269703741
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22, Problem 22E
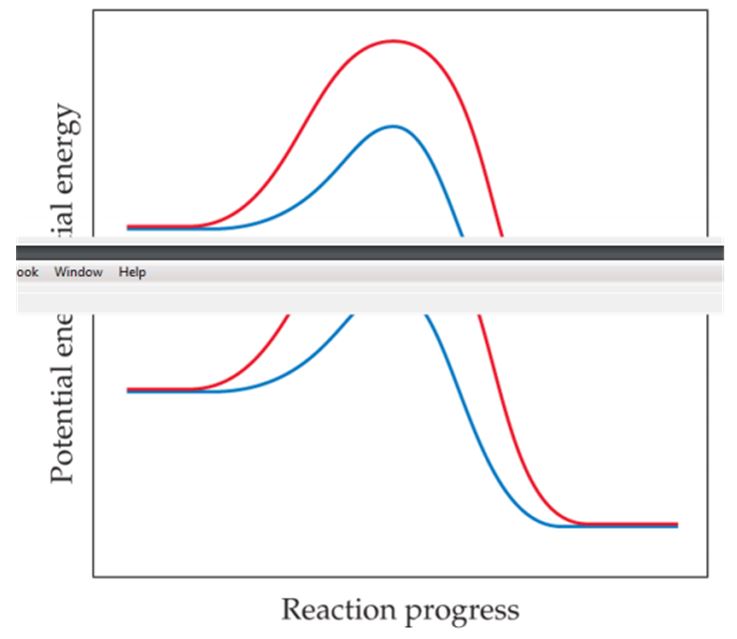
The following graph shows two different reaction pathways for the same overall reaction at the same temperature. Is each of the following statements true or false? (a) The rate is faster for the red path than for the blue path. (b) For both paths, the rate of the reverse reaction is slower than the rate of the forward reaction. (c) The energy change ΔE is the same for both paths. [Section 14.6]
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2a) explain the difference between the net energy change of a reaction and the activation energy of the same reaction
In the following chemical reaction
A + B → 2C + 40KJ
at a certain temperature, if you know that the potential energy of the reactants is 80 kilojoules, and the potential energy of the activated complex is 160 kilojoules, and when a catalyst is added, the activation energy for the forward reaction becomes 50 kilojoules,
a. What is the value of the activation energy for a reverse reaction in kilojoules in the presence of the catalyst? And without the catalyst?
b. What is the value of the potential energy (in kilojoules) of the products of a chemical reaction?
c. What is the value of the activated Complex potential energy (in kilojoules) in the presence of the catalyst?
7.For a chemical reaction, the activation energy for the forward reaction is +181 kJ and the activation energyfor the backward reaction is +62 kJ. What is the overall energy change for the forward reaction?a. –119 kJb. –62 kJc. +119 kJd. +181 kJe. +243 kJ
Chapter 22 Solutions
Chemistry: Central Science - Package (Custom)
Ch. 22.1 - Prob. 21.1.1PECh. 22.1 - Prob. 21.1.2PECh. 22.1 - Prob. 21.2.1PECh. 22.1 - Prob. 21.2.2PECh. 22.3 - At 25 ° C, the decomposition of dinitrogen...Ch. 22.3 - Practice Exercise 2 The decomposition of dimethyl...Ch. 22.4 - Practice Exercise 1 For a certain reaction A ...Ch. 22.4 - Prob. 21.4.2PECh. 22.7 - Prob. 21.7.1PECh. 22.7 - Prob. 21.7.2PE
Ch. 22.10 - Prob. 21.10.1PECh. 22.10 - Prob. 21.10.2PECh. 22.10 - Prob. 21.7.1PECh. 22.10 - Prob. 21.7.2PECh. 22 - Prob. 1DECh. 22 - Prob. 1ECh. 22 - Prob. 2ECh. 22 - Prob. 3ECh. 22 - Prob. 4ECh. 22 - The gas-phase reaction CL (g) + HBr (g) + HCl (g)...Ch. 22 - What is the molecularity of each of the following...Ch. 22 - Prob. 7ECh. 22 - Prob. 8ECh. 22 - Cyclopentadiene (C5H6) reacts with itself to form...Ch. 22 - Practice Exercise 1 An Alternative two-step...Ch. 22 - Prob. 11ECh. 22 - Practice Exercise 1
Consider the...Ch. 22 - Prob. 13ECh. 22 - Prob. 14ECh. 22 - Prob. 15ECh. 22 - Prob. 16ECh. 22 - You study the rate of a reaction, measuring both...Ch. 22 - Suppose that for the reaction K+L M, you monitor...Ch. 22 - Prob. 19ECh. 22 - Prob. 20ECh. 22 - Prob. 21ECh. 22 - The following graph shows two different reaction...Ch. 22 - Prob. 23ECh. 22 - Prob. 24ECh. 22 - Prob. 25ECh. 22 - Prob. 26ECh. 22 - Prob. 27ECh. 22 - Prob. 28ECh. 22 - Prob. 29ECh. 22 - Prob. 30ECh. 22 - Prob. 31ECh. 22 - Prob. 32ECh. 22 - Prob. 33ECh. 22 - Prob. 34ECh. 22 - Prob. 35ECh. 22 - Prob. 36ECh. 22 - Prob. 37ECh. 22 - Prob. 38ECh. 22 - Prob. 39ECh. 22 - Prob. 40ECh. 22 - Prob. 41ECh. 22 - Prob. 42ECh. 22 - Prob. 43ECh. 22 - Prob. 44ECh. 22 - Prob. 45ECh. 22 - Prob. 46ECh. 22 - Prob. 47ECh. 22 - Prob. 48ECh. 22 - Prob. 49ECh. 22 - Prob. 50ECh. 22 - Prob. 51ECh. 22 - Prob. 52ECh. 22 - Prob. 53ECh. 22 - Prob. 54ECh. 22 - Prob. 55ECh. 22 - Prob. 56ECh. 22 - Prob. 57ECh. 22 - Prob. 58ECh. 22 - Prob. 59ECh. 22 - Prob. 60ECh. 22 - Prob. 61ECh. 22 - Prob. 62ECh. 22 - Prob. 63ECh. 22 - Prob. 64ECh. 22 - Prob. 65ECh. 22 - Prob. 66ECh. 22 - Prob. 67ECh. 22 - Prob. 68ECh. 22 - Prob. 69ECh. 22 - Prob. 70ECh. 22 - Prob. 71ECh. 22 - Prob. 72ECh. 22 - Prob. 73ECh. 22 - Prob. 74ECh. 22 - Prob. 75ECh. 22 - Prob. 76ECh. 22 - Prob. 77ECh. 22 - Prob. 78ECh. 22 - Prob. 79ECh. 22 - Prob. 80ECh. 22 - Prob. 81AECh. 22 - Prob. 82AECh. 22 - Prob. 83AECh. 22 - Prob. 84AECh. 22 - Prob. 85AECh. 22 - Prob. 86AECh. 22 - Prob. 87AECh. 22 - Prob. 88AECh. 22 - Prob. 89AECh. 22 - Prob. 90AECh. 22 - Prob. 91AECh. 22 - Prob. 92IECh. 22 - Prob. 93IECh. 22 - Prob. 94IECh. 22 - Prob. 95IECh. 22 - Prob. 96IECh. 22 - Prob. 97IECh. 22 - Prob. 98IECh. 22 - Prob. 99IECh. 22 - Prob. 100IECh. 22 - Prob. 101IECh. 22 - Prob. 102IECh. 22 - Prob. 103IECh. 22 - Prob. 104IECh. 22 - Prob. 105IECh. 22 - Prob. 106IECh. 22 - Prob. 107IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- . What does the activation energy for a reaction represent? How is the activation energy related to whether a collision between molecules is successful?arrow_forward(a) Explain the following terms :(i) Order of a reaction(ii) Molecularity of a reaction(b) The rate of a reaction increases four times when the temperature changes from 300 K to 320 K. Calculate the energy of activation of the reaction, assuming that it does not change with temperature. (R = 8.314 J K-1 mol-1)arrow_forwardWhich of the following does not affect the rate of a chemical reaction? Group of answer choices A) The equilibrium constant of the reaction. B) The concentration of reactants unless the reaction is zero-order to any reactant. C) The temperature. D) The activation energy of the reaction. E) The presence of a catalyst.arrow_forward
- Which of the following parameters would be different for a reaction carried out in the presence of a catalyst compared with the same reaction carried out in the absence of a catalyst? G°, H‡, Ea, S‡, H°, Keq, G‡, S°, karrow_forwardA student constructed in the rainiest place in found a line of best fit of y=-6,627x+1,532. What is the energy of activation (in kJ/mol).arrow_forwardb. Calculate the rate constant, with proper units, for the forward reaction c What is the complete rate law for the forward reaction? d. If the rate constant for the reverse reaction is 9.3 x 10 10 , what is Kc for the reactionarrow_forward
- Given the Equation: 2C(g) + 2D(g) = 3E(g) + F(g) + heat ,Predict the direction of the shift in equilibrium, Addition of a catalyst.a. forwardb. backwardc. no changeMost reactions are carried out in liquid solution or in the gaseous phase because in such situationsa. Activation energies are higher.b. It is easier for reactants to come in contact with each other.c. Kinetic energies of reactants are lower.d. Products are less apt to decompose. Whether a reaction is exothermic or endothermic is determined bya. the activation energyb. the physical state of the reactantsc. how reactant and product potential energies compared. whether a catalyst is presente. None Which of the following changes is most likely to decrease reaction rate for the chemical reaction 2CO + O2 ---> 2CO2?a. adding twice as much O2 to the reaction mixtureb. tripling the temperature of the reaction mixturec. adding a catalyst to the reaction mixtured. removing some CO from the reaction mixturee. Nonearrow_forwardDefine the following terms.(i) Rate constant (k) (ii) Activation energy ( Ea)arrow_forwardIf reaction A has an activation energy of 250 kJ/mol and reaction B has an activation energy of 100 kJ/mol, which of the following statements must be correct? If reaction A is exothermic and reaction B is endothermic then reaction A is favored kinetically. The energy of reaction B must be greater than the energy of reaction A. At the same temperature the rate of reaction B is greater than the rate of reaction A. The rate of reaction A at 25 °C equals the rate of reaction B at 100 °C. The energy of reaction A must be greater than the energy of reaction B.arrow_forward
- Which of the following statements is true? a. The concentration of a catalysts steadily decreases as a reaction proceeds b. A catalyst functions by selectively retarding the reverse directions c. A catalyst functions by lowering the activation energy for a reaction. d. A catalyst changes the ∆H for the reaction.arrow_forwardSketch the energy graphs for the following situations a. The reactants have a lower potential energy than the products. b. The activation energy of the reverse reaction is greater than the forward reaction. c. The products have a lower potential energy than the reactants. d. The forward reaction has a positive delta H. e. The reverse reaction has a negative delta H.arrow_forward4B(s) + 3O2(g) → 2B2O3(s) ∆HO= -2543.8 kJ Calculate the ∆HO for the decomposition of B2H6into its elements.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY