
Concept explainers
Synthetic Applications of Enamines
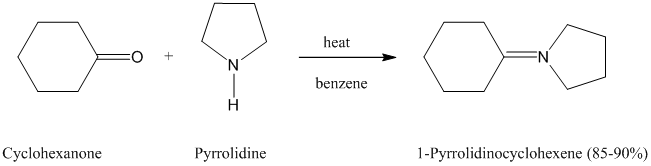
The formation of enamines by the reaction of
described in Section

When preparing enamines, the reaction is normally carried out by heating in benzene as the solvent. No catalyst is necessary, but
Enamines resemble enols in that electron-pair donation makes their double bond electron-rich
and nucleophilic.
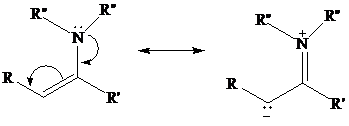
Because nitrogen is a better electron-pair donor than oxygen, an enamine is more nucleophilic thanan enol. Enamines, being neutral molecules, are, however, less nucleophilic than enolates, which areanions.
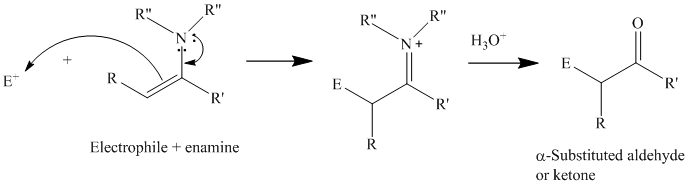
Reactions of enamines with electrophiles
Pyrrolidine is the secondary amine used most often for making enamines from aldehydes and
ketones.
For synthetic purposes, the electrophilic reagents that give the best yields of
Acyl chlorides and acid anhydrides.
Michael acceptors:
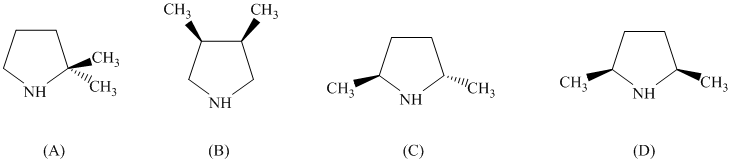
pyrrolidine derivative. Of the following, which one is the best pyrrolidine derivative to use
in this enantioselective synthesis?
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
ORGANIC CHEMISTRY (LL)-W/SOLN.>CUSTOM<
- How can ferric chloride reagent be use to test for basicity of amines? Provide the chemical equation for this reaction.arrow_forwardDescribe the product formed as a result of the reaction between benzylamine and ethylacrylate by writing the mechanism of the reaction.arrow_forwardGive a chemical test to distinguish between each of the following pairs of compounds :(i) Ethylamine and Aniline(ii) Aniline and Benzylaminearrow_forward
- Salsalate, which is an ester formed by the reaction of two molecules of salicylic acid, is another salicylate used as an aspirin alternative for those who are hypersensitive to aspirin. Draw the structures of salicylic acid and salsalate.arrow_forward2. Write the equation for the reaction of acetanilide with molecular bromine (for the organic compounds, draw the full structures, not condensed formulae).arrow_forwardQ4:- Draw structures, give names, and classify as primary, secondary, or tertiary amine : the eight isomeric amines of formula CaHi.N Q5:- Give structures and names of the principal organic reactants and products of the following reactions. a- conversion of Carboxylic Acids into Esters b- Hydrolysis of nitrites. c- Hofmann degradation of amides Q6:- Arrange the compounds of each set in order of basicity ? Explain your choice. ethylamine, 2-aminoethanol, 3-amino-l-propanolarrow_forward
- N-Methylpyrrolidine has a boiling point of 81 °C, and piperidine has a boiling point of 106 °C. Tetrahydropyran has a boiling point of 88 °C, and cyclopentanone has a boiling point of 141 °C. These two isomershave a boiling point difference of 53 °C. Explain why the two oxygen-containing isomers have a much larger boilingpoint difference than the two amine isomersarrow_forwardAmines with more than 6 carbons are soluble in: a) aqueous HCI b) aqueous NaHCO3 d) water c) aqueous NaOH Which of the following would give a positive iodoform test? acetone a) benzophenone c) 3-pentanone d) cyclopentanone meth "Saponification" as the term is used in organic chemistry means: a) acidic hydrolysis of an ester b) basic hydrolysis of an ester c) acidic hydrolysis of an amide d) basic hydrolysis of an amide 3. ( Propylamine can be synthesized by the LiAlH4 reduction of: a) CH3CH2CECH b) CH3CH=NH d)) CH3CH2CEN c) CH3CH2NO2arrow_forwardAnilines can be converted into diazonium salts by reaction with nitrous acid. Diazonium salts react with phenols to form azo dyes. Sketch the aniline derivative and phenol reactant that can combine to produce the azo dye shown.arrow_forward
- Why is methylamine more basic than aniline?arrow_forwardEthylamine is soluble in water, whereas aniline is almost insoluble, why?arrow_forwardTrue or False Amine salts have increased solubility in water. Amines have a pleasant floral aroma. Carboxylic acids have significantly higher boiling points than alcohols of comparable molecular weight Butanal would have a higher boiling point than butanoic acid. Pentanone would have a lower boiling point than pentane. Carboxylate ions have decreased solubility in water.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



