
The IUPAC rules permit the use of common names for a number of familiar phenols and aryl ethers. These common names are listed here along with their systematic names. Write the structure of each compound.
(a)
(b)
(c)
(d)
(e)
(f)
Interpretation:
The structures of the given compounds are to be drawn.
Concept introduction:
The systematic naming of organic compound is done by using IUPAC nomenclature. The naming of an organic compound is done in such a way that the structure of the organic compound is correctly interpreted from its name.
In order to determine the structure of an organic compound from its name, first the root word in the name is identified. The suffixes like
In the next step of structure identification of the organic compound from its name, the position, location and number of the substituents bonded to the carbon chain are determined.
Answer to Problem 12P
Solution:
a) The structure of
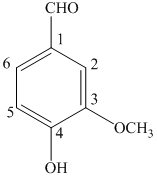
b) The structure of
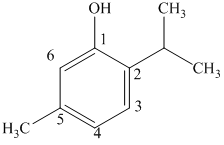
c) The structure of
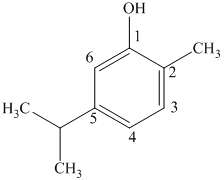
d) The structure of
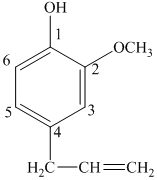
e) The structure of
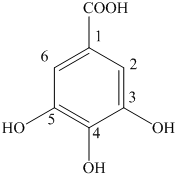
f) The structure of
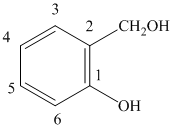
Explanation of Solution
a) The structure of
The given compound is

b) The structure of
The given compound is

c) The structure of
The given compound is

d) The structure of
The given compound is

e) The structure of
The given compound is

f) The structure of is
The given compound is

Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY-PACKAGE >CUSTOM<
- Provide the structure of the major organic product of the following reaction and? explain the stereochemistry which results in this product. 2-Pentanol reacting with 1.) PBr3, pyridine 2.) NaCNarrow_forwardCompounds A, B, and C have the same molecular formula C4H8. They all react wotu H2/PtO2 to give the same compound. The reaction of A or B with H2O/H2SO4 or with BH3-THF, followed by treatment with a basic solution of hydrogen peroxide, gives the same compound, namely D. The reaction of C with H2O/H2SO4 also gives D. However, the reaction of C with BH3-THF, followed by HO-, H2O2 gives a new compound, E. Provide the identity of A, B, C, D, and E along with explanations of reactivity.arrow_forwardAlcohol A (C10H18O) is converted to a mixture of alkenes B and C on being heated with potassium hydrogen sulfate (KHSO4). Catalytic hydrogenation of B and C yields the same product. Assuming that dehydration of alcohol A proceeds without rearrangement, deduce the structures of alcohol A and alkene C.arrow_forward
- Benzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.arrow_forwardMost alkyl bromide are water-insoluble liquids. But when the following alkyl bromide Awas first isolated, its high melting point of 203oC (solid at room temperature) and its water solubility led its discoverers to comment that it behaves more like a salt (hint: like NaCl). Explain the salt-like behavior of compound A in under thirty words.arrow_forwardStearolic acid, C18H32O2, yields stearic acid on catalytic hydrogenation and undergoes oxidative cleavage with ozone to yield nonanoic acid and nonanedioic acid. What is the structure of stearolic acid?arrow_forward
- The reaction of 3,4-dimethyl-3-hexanol (3,4-dimethylhexan-3-ol) with HBr generates compound A as the major product. Treatment of compound A with a strong base gives two isomers of compound B as the major product, along with one isomer of compound C and one isomer of compound D as minor products, all of which have one double-bond equivalent. Identify compounds A, B, C, and D and give their names. By what mechanism does the reaction of 3,4-dimethyl-3-hexanol with HBr occur? By what mechanism does the reaction of A with strong base to form B occur? Propose reaction conditions for an alternative, one-step method for converting 3,4-dimethyl-3-hexanol directly to compound B.arrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questionsA)What is the IUPAC name for Compound X?B)What type of reaction (s) is/are represented by (i) and (ii)?C)Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B]. D)Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed.E)Which of the compounds in the following pairs will occur in relatively higher yields and why?I)[1] and [2] II)[3] and [4]arrow_forwardExplain the following behaviours :(i) Alcohols are more soluble in water than the hydrocarbons of comparable molecular masses.(ii) Ortho-nitrophenol is more acidic than ortho-methoxyphenol.arrow_forward
- Treatment of cis-4-bromocyclohexanol with HO– affords compound A and cyclohex-3-en-1-ol. Treatment of trans-4- bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and B contain different functional groups and are not isomers of each other. Propose structures for A and B and offer an explanation for their formation.arrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. A. What is the IUPAC name for Compound X? B. What type of reaction (s) is/are represented by (i) and (ii)? C. Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B]. D. Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed. E. Which of the compounds in the following pairs will occur in relatively higher yields and why? [1] and [2] [3] and [4] The attached image contains the scheme.arrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. –a. What is the IUPAC name for Compound X? b. What type of reaction (s) is/are represented by (i) and (ii)? c. Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B]. d. Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed.e. Which of the compounds in the following pairs will occur in relatively higher yields and why?i. [1] and [2] ii. [3] and [4]arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





