
Concept explainers
How does the metal ion in carboxypeptidase A increase the enzyme’s catalytic activity
Interpretation:
The metal ion in carboxypetidase A increases the catalytic activity of the enzyme; this has to be explained.
Explanation of Solution
Carboxypetidase A is a digestive enzyme. It hydrolyzes the carboxyl terminal peptide bond and it prefers bulky and aliphatic residues.
Proposed mechanism for the catalytic reaction of carboxypetidase is given in figure 1.
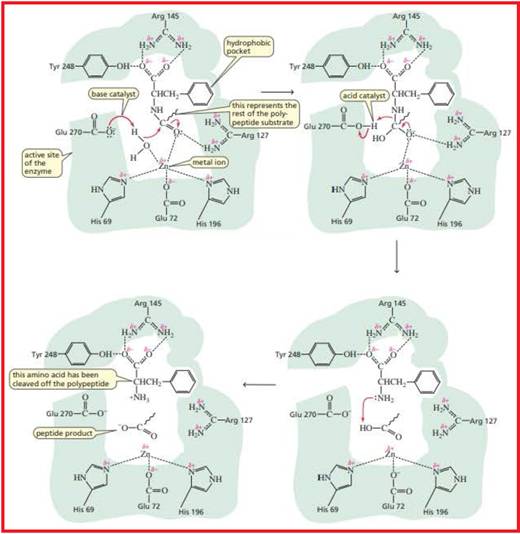
Figure 1
The metal ion present in the carboxypeptidase A enzyme is Zinc metal.
This metal ion
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEM ALC PKG
- In the multienzyme sequence shown below, molecules of D are able to fit to the enzyme E1 and prevent the conversion of A to B. What is this action of E called? AE1BE2CE3D a. effector inhibition b. allosteric inhibition c. feedback inhibition d. competitive inhibition by nonproductarrow_forwardDraw a graph that shows the effect of increasing substrate concentration on the rate of an enzyme-catalyzed reaction (at constant temperature, pH, and enzyme concentration).arrow_forwardExplain how the pasteurization of milk utilizes one of the factors that influence enzyme activity.arrow_forward
- The site on an enzyme molecule that does the catalytic work is called the: a. binding site. b. allosteric site. c. lock. d. active site.arrow_forwardDescribe the importance of zymogens in the body. Give an example of an enzyme that has a zymogen.arrow_forwardDescribe the difference between graphs showing temperature versus reaction rate for an enzyme-catalyzed reaction and an uncatalyzed reaction.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





