
Concept explainers
Interpretation:
The condensed structure and possible isomers should be drawn and identified for the given molecular model structure.
Concept Introduction:
Condensed Structure: It is the one, where the symbols of atoms are listed in order as they appear in the molecules structure with bond dashes omitted or limited.
- 1. A molecular formula explains only the number of atoms of each element in a molecule of the compound.
- 2. Condensed structure shows all atoms, but it omits the vertical bonds and most or all of the vertical bonds and most or all of the horizontal single bonds.
Isomers: The molecules with same molecular formula but differ in the way in which the atoms are arranged is known as isomers.
In other words compounds have the same molecular formula but have different structural formula and different names.
Answer to Problem 23.33CP
Condensed structure of given molecular model is,
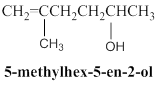
Explanation of Solution
Given molecular model presence of seven carbon atoms and 14 hydrogen atoms and all hydrogen atoms horizontally connected to respective carbon atoms, so given molecular model is correctly matched for a 5-methylhex-5-ene-2-ol molecule and condensed structure is shown below.
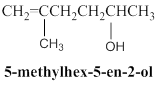
Determination of possible isomers:
Above the condensed structure was found to have two possible isomers, the possible isomers structure are shown below,
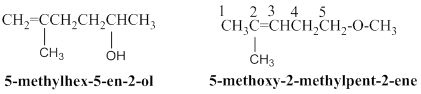
5-methyl hex-5-en-2-ol:
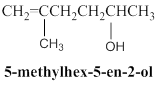
The above structure, 11 hydrogen atoms are horizontally connected to respective carbon atoms, one methyl groups is connected in branched position and another one hydroxyl group connected in
5-methoxy-2-methylpent-2-ene:
Given molecular model structure was founded to have another possible isomers,
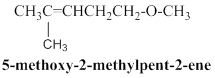
In second molecule 11 hydrogen atoms are horizontally connected to respective carbon atoms, and one methyl group is connected in branched position, than another one methoxy group connected in
For the given molecular model, the condensed structure was drawn and possible isomers were determined.
Want to see more full solutions like this?
Chapter 23 Solutions
General Chemistry: Atoms First -Laboratory Manual
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




