
(a)
Interpretation: The
Concept introduction: The Malonic ester synthesis is a method which is used to convert diethyl malonate into a carboxylic acid.
Answer to Problem 23.43P
The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
Explanation of Solution
The given carboxylic acid is,
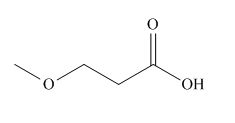
Figure 1
All alkyl groups on the alpha carbon will be obtained from alkyl halides and the remaining molecule is obtained from ethyl acetoacetate. Thus, for the required alkyl halide, the location of alpha carbon is shown below.
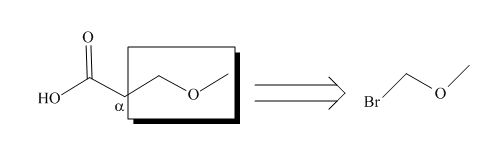
Figure 2
Hence, the alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
The reaction of diethylmalonate with
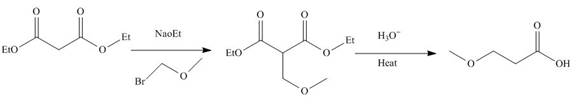
Figure 3
The mechanism involved in the given reaction as follows,
Base abstracts the proton from the alpha carbon and enolate ion reacts with
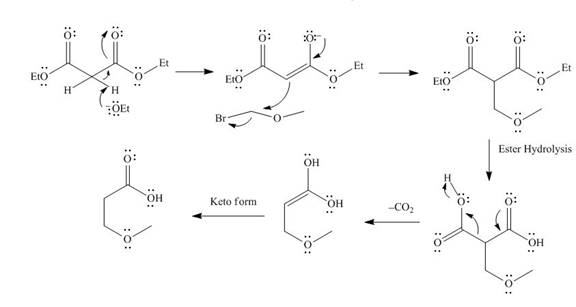
Figure 4
The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
(b)
Interpretation: The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is to be predicted.
Concept introduction: The Malonic ester synthesis is a method which is used to convert diethyl malonate into a carboxylic acid.
Answer to Problem 23.43P
The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
Explanation of Solution
The given carboxylic acid is,
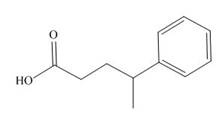
Figure 5
All alkyl groups on the alpha carbon will be obtained from alkyl halides and the remaining molecule is obtained from ethyl acetoacetate. Thus, for the required alkyl halide, the location of alpha carbon is shown below.
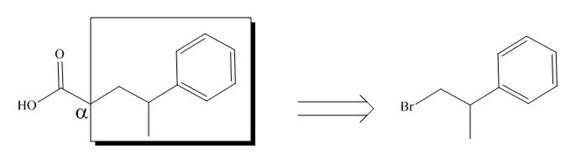
Figure 6
Hence, the alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
The reaction of diethylmalonate with
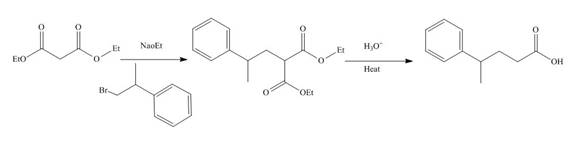
Figure 7
The mechanism involved in the given reaction as follows,
Base abstracts the proton from the alpha carbon and enolate reacts with
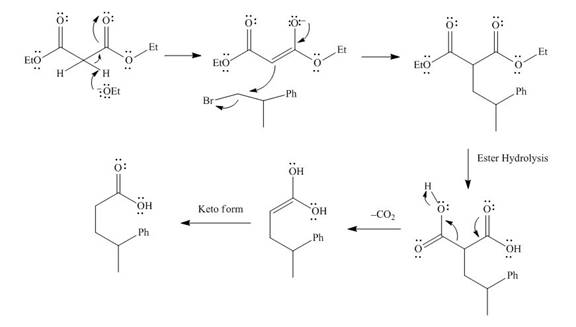
Figure 8
The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
(c)
Interpretation: The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is to be predicted.
Concept introduction: The Malonic ester synthesis is a method which is used to convert diethyl malonate into a carboxylic acid.
Answer to Problem 23.43P
The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
Explanation of Solution
The given carboxylic acid is,
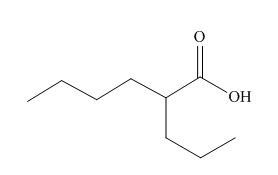
Figure 9
All alkyl groups on the alpha carbon will be obtained from alkyl halides and the remaining molecule is obtained from ethyl acetoacetate. Thus, for the required alkyl halide, the location of alpha carbon is shown below.
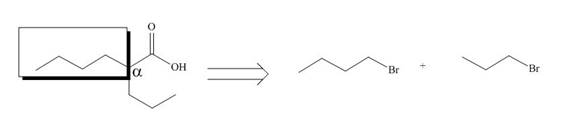
Figure 10
Hence, the alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
The reaction of diethylmalonate with
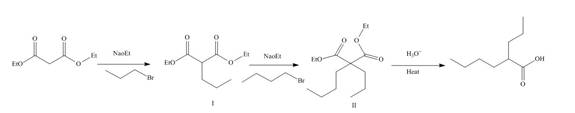
Figure 11
The mechanism involved in the given reaction as follows,
Base abstracts the proton from the alpha carbon and enolate reacts with
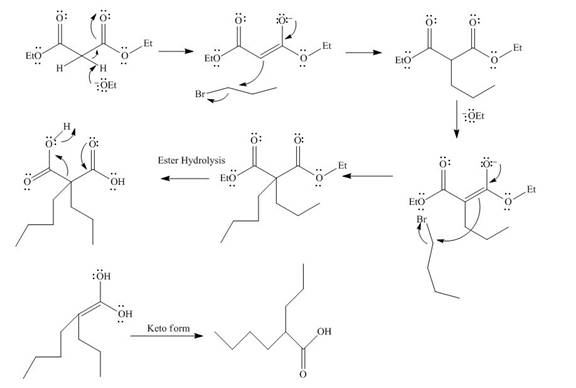
Figure 12
The alkyl halide which is needed to prepare given carboxylic acid using the malonic ester synthesis is
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEM.(LL)W/STD GDE.+CONNECT PKG
- Devise a synthesis of each compound from phenol (C6H5OH) and any other organic or inorganic reagents.arrow_forwardDevise a synthesis of each compound from aniline (C6H5NH2) as starting material.arrow_forwardWhat product is formed when a solution of A and B is treated with mild base? This reaction is the rst step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forward
- Carboxylic acid X is an intermediate in the multistep synthesis ofproparacaine, a local anesthetic. Devise a synthesis of X from phenoland any needed organic or inorganic reagents.arrow_forwardDraw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that can be used as a synthetic detergent to clean away dirt (see Problem 3.22).arrow_forwardDraw a stepwise mechanism for the attached reaction, which involves two Friedel–Crafts reactions. B was an intermediate in the synthesis of the antidepressant sertralinearrow_forward
- Carboxylic acid X is an intermediate in the multistep synthesis of proparacaine, a local anesthetic. Devise a synthesis of X from phenol and any needed organic or inorganic reagents.arrow_forwardDevise a synthesis of attached compound. You may use HC=CH, ethyleneoxide, and alkyl halides as organic starting materials and any inorganicreagents.arrow_forwardOne synthesis of the cholesterol-lowering drug atorvastatin involves the construction of the pyrrole by reaction of diketone X with amine Y. Draw a stepwise mechanism for this reaction.arrow_forward
- Draw a stepwise mechanism for the following reactions, two steps in R. B. Woodward’s classic synthesis of reserpine in 1958. Reserpine, which is isolated from the extracts of the Indian snakeroot Rauwola serpentina Benth, was used at one time to manage mild hypertension associated with anxiety.arrow_forwardDraw a stepwise mechanism for the following reactions, two steps in R. B. Woodward’s classic synthesis of reserpine in 1958. Reserpine, which is isolated from the extracts of the Indian snakeroot Rauwolfia serpentina Benth, was used at one time to manage mild hypertension associated with anxiety.arrow_forwardDraw a stepwise mechanism for the following reaction of a Grignard reagent with a cyclic amide.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





