
(a)
Interpretation: The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
Concept introduction: The replacement or substitution of one functional group with another different functional group in any
The bulky base like LDA always abstracts the proton from the side of less substituted
Answer to Problem 23.7P
The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
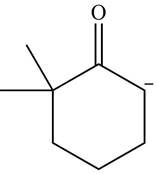
Explanation of Solution
The product that is formed by the reaction of the given starting material with LDA in THF solution at
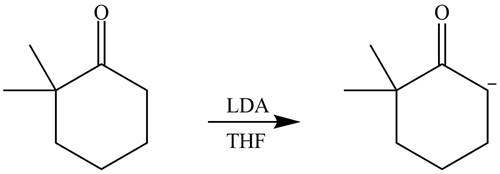
Figure 1
In this reaction,
(b)
Interpretation: The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
The bulky base like LDA always abstracts the proton from the side of less substituted
Answer to Problem 23.7P
The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
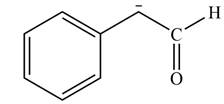
Explanation of Solution
The product that is formed by the reaction of the given starting material with LDA in THF solution at
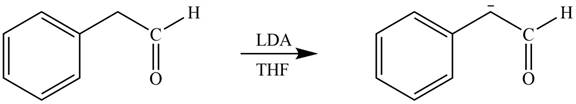
Figure 2
In this reaction,
(c)
Interpretation: The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
The bulky base like LDA always abstracts the proton from the side of less substituted
Answer to Problem 23.7P
The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
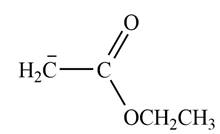
Explanation of Solution
The product that is formed by the reaction of the given starting material with LDA in THF solution at
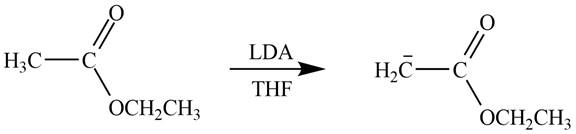
Figure 3
In this reaction, ethyl acetate is treated with LDA in the presence of THF solution to form an enolate ion. The strong base, LDA abstracts a proton from the less substituted carbon atom of the compound.
(d)
Interpretation: The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
The bulky base like LDA always abstracts the proton from the side of less substituted
Answer to Problem 23.7P
The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
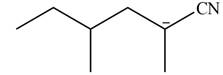
Explanation of Solution
The product that is formed by the reaction of the given starting material with LDA in THF solution at
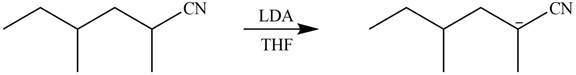
Figure 4
In this reaction,
(a) The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
(b) The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
(c) The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
(d) The product that is formed by the reaction of the given starting material with LDA in the presence of THF solution at
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY LSLF W/CONNECT >BI<
- Draw the products formed when each dihalide is treated with excess NaNH2.arrow_forwardDraw the products formed when each dihalide is treated with excessNaNH2.arrow_forwardDraw the products formed when A is treated with each reagent: (a) H2 + Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H3SO4, H2O; (e) Sharpless reagent with (+)-DET.arrow_forward
- Draw the products formed from nitration of each compoundarrow_forwardDraw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs.a. NaBH4, CH3OHb. [1] LiAlH4; [2] H2Oc. [1] CH3MgBr (excess); [2] H2Od. [1] C6H5Li (excess); [2] H2Oe. Na2Cr2O7, H2SO4, H2Oarrow_forwardEthers are oxidized with O2 to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.arrow_forward
- (plz with detaol explanation )arrow_forward(a) Give an acceptable name for each compound, (b) Draw the organic products formed when A or B is treated with each reagent: [1] H3O+; [2] −OH, H2O; [3] CH3CH2CH2MgBr (excess), then H2O; [4] LiAlH4, then H2O.arrow_forwardDraw a stepwise mechanism for the conversion of dihalide A and cyclohexane-1,4-diol to polyether B in the presence of AlCl3.arrow_forward
- Nicotine can be made when the attached ammonium salt is treated withNa2CO3. Draw a stepwise mechanism for this reaction.arrow_forwardWhat Wittig reagent and carbonyl compound are needed to prepare each alkene? When two routes are possible, indicate which route, if any, is preferred.arrow_forwardDraw the products formed when each compound is treated with HNO3 and H2SO4.State whether the reaction occurs faster or slower than a similar reaction with benzene.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





