
a.
Interpretation: To write the expected products that are obtained by the following reactions.
Concept Introduction: Addition reactions are those in which two or more reactants combine together to form a product. The addition of hydrogen and halogen will follow Markonikov’s rule. In the case of water,
a.
Answer to Problem 39A
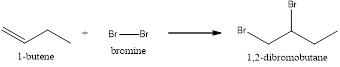
Explanation of Solution
In this reaction, the pi of the alkene and the sigma bond of the alkene will break resulting in the formation of a new bond. This reaction will follow Markonikov’s rule. In the first step, the double bond of the alkene breaks and results in the formation of a carbocation. In the second step, the bond between the halogen breaks and gets attached to the carbocation. Now the halogen will get attached to the carbocation resulting in the formation of 1,2-dibromobutane.
b.
Interpretation: To write the expected products that are obtained by the following reactions.
Concept Introduction: Addition reactions are those in which two or more reactants combine together to form a product. The addition of hydrogen and halogen will follow Markonikov’s rule. In the case of water, alkenes undergo an addition reaction in the presence of a catalyst resulting in the formation of alcohols.
b.
Answer to Problem 39A

Explanation of Solution
In this reaction, the pi of the alkene and the sigma bond of the alkene will break resulting in the formation of a new bond. This reaction will follow Markonikov’s rule. In the first step, the double bond of the alkene breaks and results in the formation of carbocation. In the second step, the bond between the halogen breaks and gets attached to the carbocation. Now the halogen will get attached to the carbocation resulting in the formation of 2,3-diiodibutane.
c.
Interpretation: To write the expected products that are obtained by the following reactions.
Concept Introduction: Addition reactions are those in which two or more reactants combine together to form a product. The addition of hydrogen and halogen will follow Markonikov’s rule. In the case of water, alkenes undergo an addition reaction in the presence of a catalyst resulting in the formation of alcohols.
c.
Answer to Problem 39A
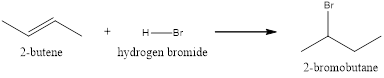
Explanation of Solution
The above reaction is an electrophilic addition reaction which is useful for the preparation of
d.
Interpretation: To write the expected products that are obtained by the following reactions.
Concept Introduction: Addition reactions are those in which two or more reactants combine together to form a product. The addition of hydrogen and halogen will follow Markonikov’s rule. In the case of water, alkenes undergo an addition reaction in the presence of a catalyst resulting in the formation of alcohols.
d.
Answer to Problem 39A
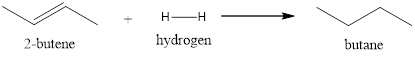
Explanation of Solution
This reaction takes place in the presence of a catalyst. The pi-bond of the alkene will act as a source of electrons for electrophiles. The bond between the hydrogen breaks in the presence of the catalyst resulting in the formation of the metal-hydrogen bond. The surface of the metal absorbs the alkene and helps in adding the other hydrogen resulting in the formation of a saturated compound.
e.
Interpretation: To write the expected products that are obtained by the following reactions.
Concept Introduction: Addition reactions are those in which two or more reactants combine together to form a product. The addition of hydrogen and halogen will follow Markonikov’s rule. In the case of water, alkenes undergo an addition reaction in the presence of a catalyst resulting in the formation of alcohols.
e.
Answer to Problem 39A
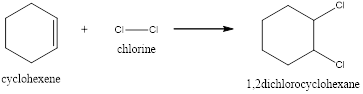
Explanation of Solution
In this reaction, the pi of the alkene and the sigma bond of the alkene will break resulting in the formation of a new bond. In the first step, the double bond of the alkene breaks and results in the formation of a carbocation. In the second step, the bond between the halogen breaks and gets attached to the carbocation. Now the halogen will get attached to the carbocation resulting in the formation of 1,2-dichlorocyclohexane.
Chapter 23 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





