
Concept explainers
Interpretation:
Identification of the given liquid is to be done and the chemical equation has to be written.
Concept introduction:
The given liquid can be either benzene or cyclohexene. The reaction of bromine vapour with an alkene gives decolorisation of bromine vapours. This reaction is used for the test of unsaturation or double bond in hydrocarbons.
Bromination of
Alkene undergoes bromination which yields the dibromo compound (vicinal dibromides or 1,2-dibromides).
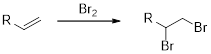
Benzene does not react with bromine vapours. However, it undergoes electrophilic substitution reaction with halogens in presence of a lewis acid such as
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry & Chemical Reactivity
- Write a balanced chemical equation that shows the formation of cyclohexane from the elements carbon (C) and hydrogen (H2 ).arrow_forwardExplain by the equation how cyclohexane can be obtained from benzene?arrow_forwardWhich alcohols can be prepared as a single product by hydroboration– oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?arrow_forward
- Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forward1. What is formed when a halogen molecule replaces hydrogen molecule(s) in an aromatic hydrocarbon? 2. What is formed when hydrogen molecules are replaced in an aliphatic hydrocarbon by halogen molecules? 3. What are a class of chemical compounds containing a hydroxyl group (-OH) bonded directly to an aromatic hydrocarbon group (like benzene)? 4. used to test for the presence of aromatic compounds in alcohols? 5. The dehydration of alcohols in the formation of ethers happens at _________.arrow_forwardWhat are the organic products of this reaction?arrow_forward
- What is the line structure and structural formula of 2-bromo-3-ethyl hexanal?arrow_forwardOzone is an oxidizing agent that cleaves a double bond and creates ________ and ________ from alkenes. ketones, carboxylic acids carboxylica acids, esters ketones, aldehydes aldehydes, estersarrow_forwardWhat reacts with benzene to produce chlorobenzene and HClarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

