
(a)
Interpretation: Via Heck reaction, the coupling partners that can be used to prepare the given compound have to be identified.
Concept Introduction:
Heck reaction:
It is the coupling reaction between an aryl, vinyl or benzyl halide and an
Examples:
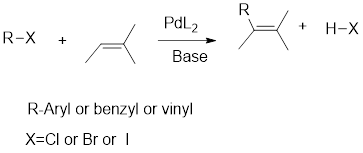
The halogen atom will be mostly iodine because it is the least electronegative atom so that it can readily leave as leaving group.
The coupling partners for the given compound can be found using the retro synthetic approach as shown here:
Example:
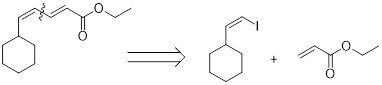
(b)
Interpretation: Via Heck reaction, the coupling partners that can be used to prepare the given compound have to be identified.
Concept Introduction:
Heck reaction:
It is the coupling reaction between an aryl, vinyl or benzyl halide and an alkene in the presence of an appropriate
Examples:

The halogen atom will be mostly iodine because it is the least electronegative atom so that it can readily leave as leaving group.
The coupling partners for the given compound can be found using the retro synthetic approach as shown here:
Example:
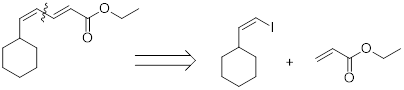
(c)
Interpretation: Via Heck reaction, the coupling partners that can be used to prepare the given compound have to be identified.
Concept Introduction:
Heck reaction:
It is the coupling reaction between an aryl, vinyl or benzyl halide and an alkene in the presence of an appropriate
Examples:

The halogen atom will be mostly iodine because it is the least electronegative atom so that it can readily leave as leaving group.
The coupling partners for the given compound can be found using the retro synthetic approach as shown here:
Example:
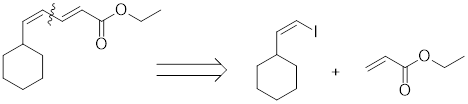
(d)
Interpretation: Via Heck reaction, the coupling partners that can be used to prepare the given compound have to be identified.
Concept Introduction:
Heck reaction:
It is the coupling reaction between an aryl, vinyl or benzyl halide and an alkene in the presence of an appropriate
Examples:

The halogen atom will be mostly iodine because it is the least electronegative atom so that it can readily leave as leaving group.
The coupling partners for the given compound can be found using the retro synthetic approach as shown here:
Example:

(e)
Interpretation: Via Heck reaction, the coupling partners that can be used to prepare the given compound have to be identified.
Concept Introduction:
Heck reaction:
It is the coupling reaction between an aryl, vinyl or benzyl halide and an alkene in the presence of an appropriate
Examples:

The halogen atom will be mostly iodine because it is the least electronegative atom so that it can readily leave as leaving group.
The coupling partners for the given compound can be found using the retro synthetic approach as shown here:
Example:

(f)
Interpretation: Via Heck reaction, the coupling partners that can be used to prepare the given compound have to be identified.
Concept Introduction:
Heck reaction:
It is the coupling reaction between an aryl, vinyl or benzyl halide and an alkene in the presence of an appropriate
Examples:

The halogen atom will be mostly iodine because it is the least electronegative atom so that it can readily leave as leaving group.
The coupling partners for the given compound can be found using the retro synthetic approach as shown here:
Example:

Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
ORGANIC CHEMISTRY..LL W/SM, SG,SM ACCES
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





