
Interpretation:
The predominant species of glutamic acid in strongly acidic and basic solutions and at its isoelectric point is to be predicted, and the higher isoelectric point of glutamine than that of glutamic acid is to be explained.
Concept introduction:
Amino acids contain both acidic (
In solid state, it exists as dipolar or zwitter ionic state, where
In aqueous state, equilibrium exists between cationic and anionic form. The predominant form depends on the pH and the nature of amino acid.
Answer to Problem 1PP
Solution:
a)
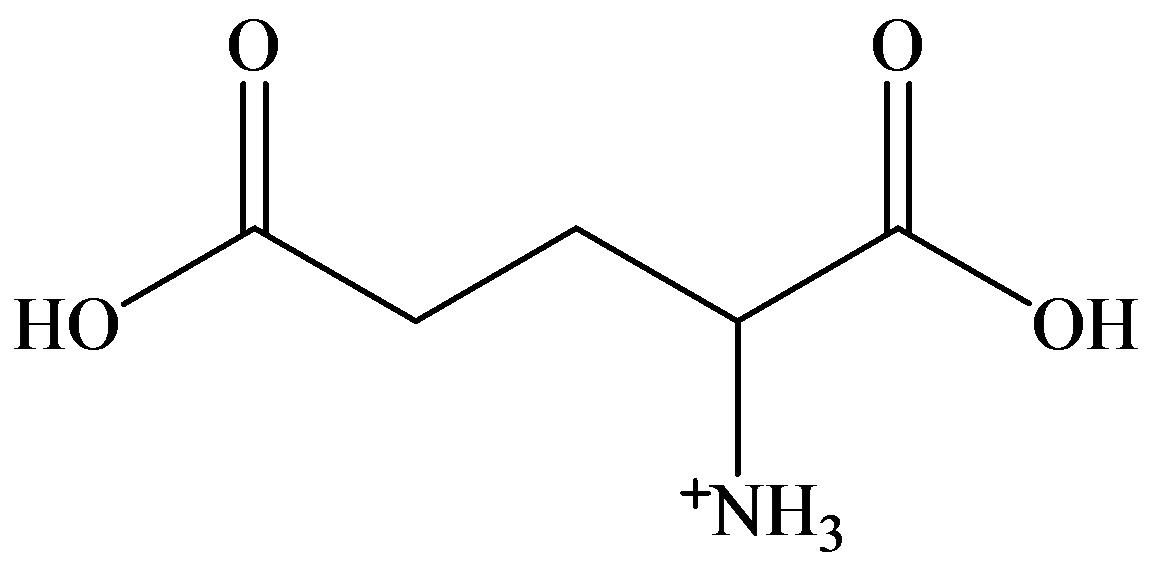
b)
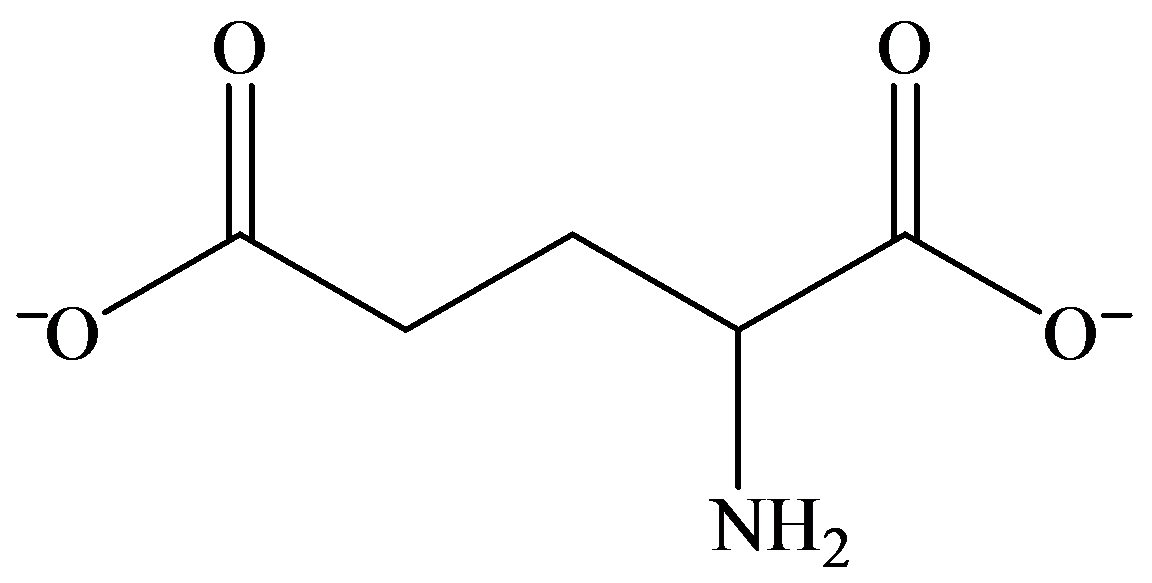
c)
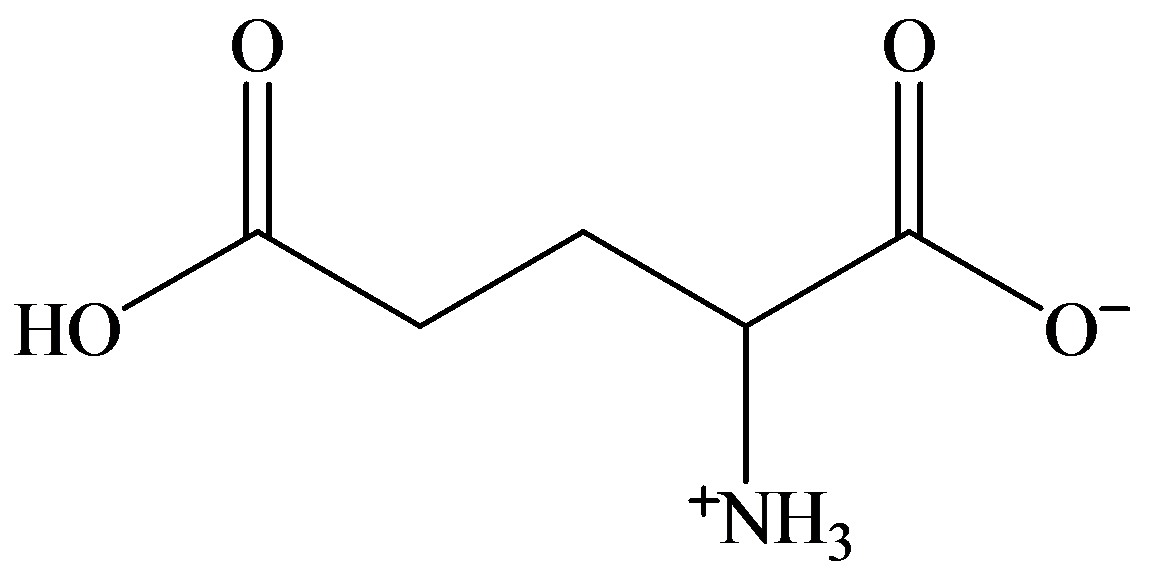
d)
Glutamic acid is a dicarboxylic acid, whereas glutamine with only one carboxyl group has a resemblance with glycine or phenylalanine. Moreover, its isoelectric point exists at a higher
Explanation of Solution
a) Strongly acidic solution
In strongly acidic solution, all amino acids are present as cations. Here,
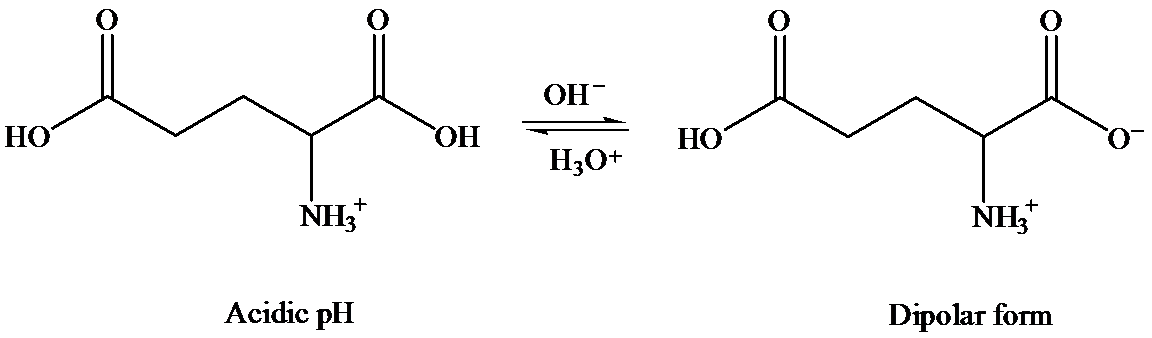
b) Strongly basic solution
In strongly basic solution, all amino acids are present as anions. The addition of base causes removal of proton from both carboxylic acid groups, resulting in a dianion. The amine group is electrically neutral.
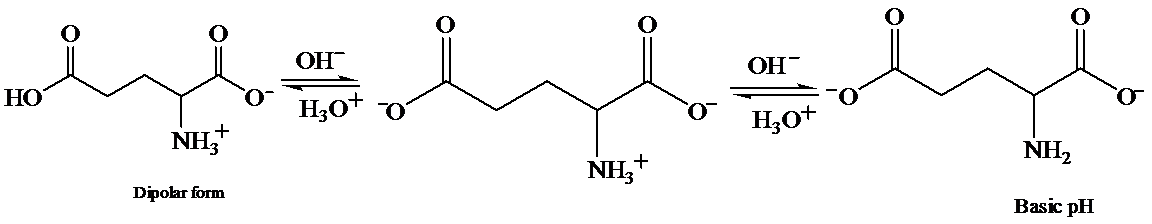
c) At the isoelectric point (pl=3.2)
Each amino acid has a particular intermediate pH at which the concentration of dipolar ion will be maximum, and the concentration of cations and anions will be equal. This point is called isoelectric point and will be different for each amino acid.
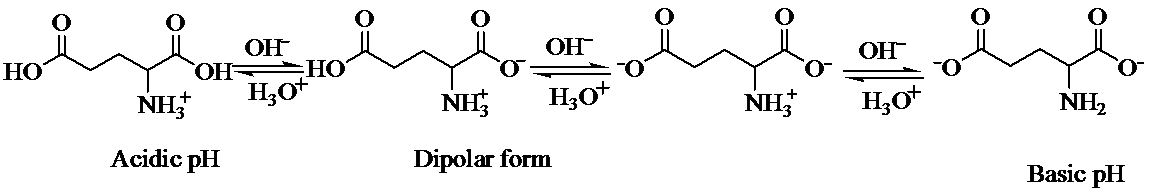
d) The isoelectric point of glutamic is consider higher than that of glutamic acid
The structure of glutamine and glutamic acid is as follows:
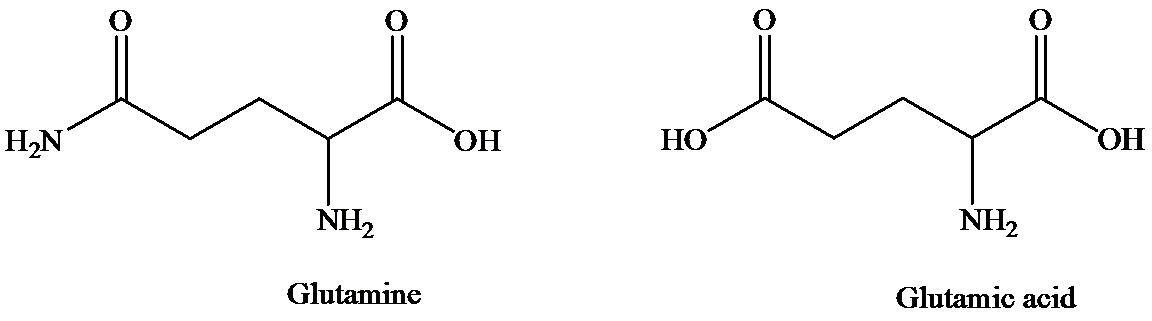
Glutamine contains only one carboxylic acid group, whereas glutamic acid has two. At isoelectric point, dipolar state exists that is there will be a carboxylate anion and an ammonium group. In order to maintain the dipolar form in glutamic acid, only one carboxylate ion is required. To suppress the ionization of second carboxylic acid, more acid is required than that for glutamine. That is pH of glutamic acid is maintained lower than that of glutamine to achieve isoelectric point. (Isoelectric point is the average of
Hence, isoelectronic point of glutamine is higher than that of glutamic acid.
Want to see more full solutions like this?
Chapter 24 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
- Phospholipids undergo saponification much like triglycerides. Draw the structure of a phospholipid meeting the followingcriteria. Then draw the products that would result from its saponification.(a) a cephalin containing stearic acid and oleic acidarrow_forward(a) The isoelectric point (pI) of phenylalanine is pH 5.5. Draw the structure of the major form of phenylalanine at pHvalues of 1, 5.5, and 11.(b) The isoelectric point of histidine is pH 7.6. Draw the structures of the major forms of histidine at pH values of 1, 4,7.6, and 11. Explain why the nitrogen in the histidine ring is a weaker base than the a-amino group.(c) The isoelectric point of glutamic acid is pH 3.2. Draw the structures of the major forms of glutamic acid at pH valuesof 1, 3.2, 7, and 11. Explain why the side-chain carboxylic acid is a weaker acid than the acid group next to thea-carbon atomarrow_forwardh) Specify the absolute (R/S) configuration of the amino group in structure IV. (i) If the substituents in structures I, IV and V were identical (all OH or all NH2), which structure would result in a meso compound? (j) If each hydroxy group for structures I, II and VI were replaced with another amino group, which compound would be made optically inactive?arrow_forward
- The first step in the catabolism of most amino acids is the removal of the nitrogen atom by transfer to an a-keto acid, a reaction catalyzed by an enzyme called a transaminase. The a-keto acid acceptor is often a-ketoglutarate. Modify the structures in the product to show the products of the transamination of cysteine. Be sure to show functional groups with the charge and number of attached hydrogen atoms appropriate for pH 7.4. transaminase + O=C H₂N-CH + CH₂ CH₂ CH₂ SH Incorrect H₂N || CH | CH₂ | CH₂ I || O || n | CH₂ T SHarrow_forwardAssign an R or S configuration to the chiral center in each amino acid. (a) l-Phenylalanine (b) l-Glutamic acid (c) l-Methioninearrow_forwardAnother method to form a peptide bond involves a two-step process Conversion of a Boc-protected amino acid to a p-nitrophenyl ester. Why does a p-nitrophenyl ester “activate” the carboxy group of thefirst amino acid to amide formation?arrow_forward
- Enumerate the appropriate methods of extraction and purification of glycosides.arrow_forwardDraw the structure of the phenylthiohydantoin derivatives of(a) alanine. (b) tryptophan. (c) lysine. (d) proline.arrow_forwardGive the polarity, melting point, boiling point, solubility, and state at room temperature of the following: Linoleic acid Tripalmitin Triolein Phosphatidylcholine(28:0) Phosphatidylinositol(34:1) Phosphatidylserine(36:2) Phosphatidylethanolamine(38:6) Cholesterolarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

