
(a)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
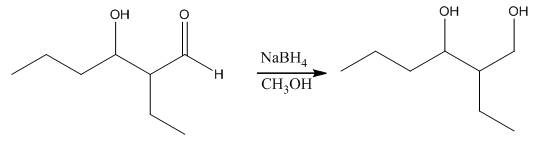
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown as,
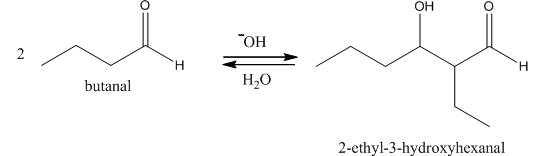
Figure 1
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert

Figure 2
Thus, the reagent that is needed to convert
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(b)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
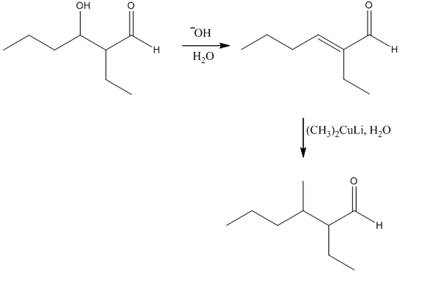
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert
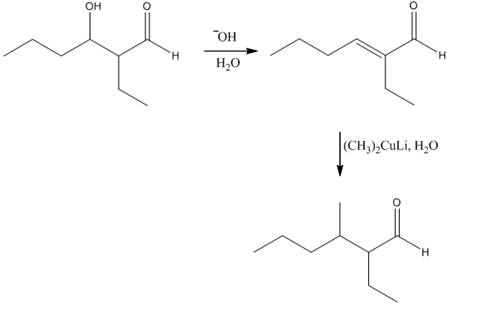
Figure 3
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(c)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
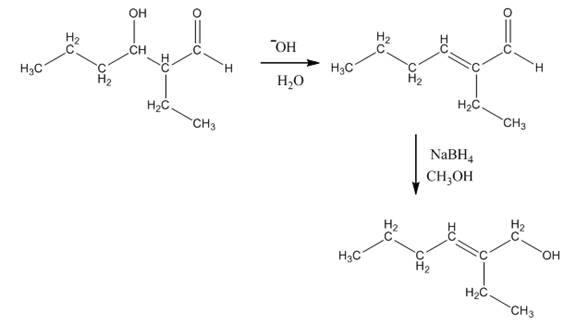
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert
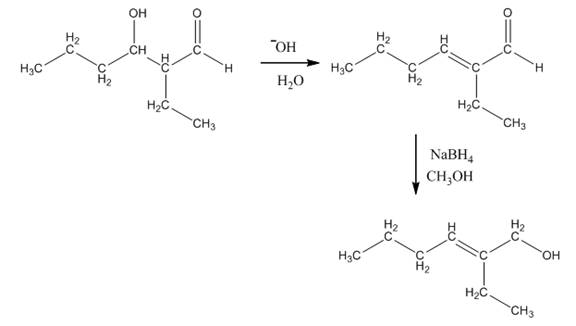
Figure 4
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(d)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
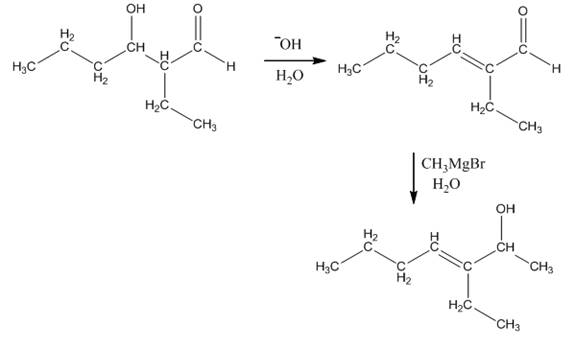
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert

Figure 5
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
Want to see more full solutions like this?
Chapter 24 Solutions
ORGANIC CHEMISTRY SOLUTION MANUAL
- Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.arrow_forwardWhat product is formed when A is treated with Grubbs catalyst underhigh-dilution conditions? This reaction was a key step in the synthesis ofstemoamide, the naturally occurring amide described in the opening paragraph.arrow_forwardDevise a stepwise mechanism for the following reaction.arrow_forward
- Identify A in the following reaction scheme, and draw a stepwise mechanism for the conversion of A to the furan B.arrow_forwardConsider carbonyl compounds A–E drawn below. (a) Rank A–E in order of increasing stability. (b) Rank A–E in order of increasing amount of hydrate formed when treated with aqueous acid. (c) Which compound is most reactive in nucleophilic addition?arrow_forwardProvide the necessary reagents next to the arrows.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


