
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24.SE, Problem 83AP
Interpretation Introduction
a) C9H13N
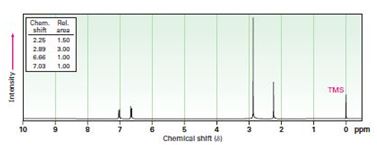
Interpretation:
To propose structures for compounds that show the following 1H NMR spectra.
Interpretation Introduction
b) C13H17N
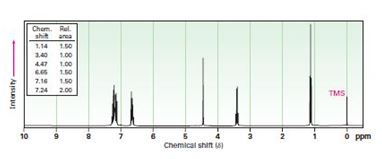
Interpretation:
To propose structures for compounds that show the following 1H NMR spectra.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose a structure for the compound, C4H8O, that has the following 1H NMR spectrum
Please propose structures for ketones or aldehydes having the following 1H NMR spectra
Propose structures for the compound C5H10O whose 1H NMR spectra is below
Chapter 24 Solutions
Organic Chemistry
Ch. 24.1 - Name the following compounds:Ch. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.3 - Prob. 4PCh. 24.3 - The benzylammonium ion (C6H5CH2NH3+) has pKa =...Ch. 24.4 - Prob. 6PCh. 24.5 - Prob. 7PCh. 24.6 - Prob. 8PCh. 24.6 - Prob. 9PCh. 24.6 - Show two methods for the synthesis of dopamine, a...
Ch. 24.6 - Prob. 11PCh. 24.6 - Prob. 12PCh. 24.6 - Prob. 13PCh. 24.7 - Prob. 14PCh. 24.7 - Prob. 15PCh. 24.8 - Prob. 16PCh. 24.8 - Propose syntheses of the following compounds from...Ch. 24.8 - How would you prepare the following compounds from...Ch. 24.8 - Prob. 19PCh. 24.9 - Draw an orbital picture of thiazole. Assume that...Ch. 24.9 - Prob. 21PCh. 24.9 - Prob. 22PCh. 24.9 - Which nitrogen atom in the hallucinogenic indole...Ch. 24.9 - Prob. 24PCh. 24.10 - Prob. 25PCh. 24.SE - Name the following amines, and identify each as...Ch. 24.SE - The following compound contains three nitrogen...Ch. 24.SE - Name the following amine, including R, S...Ch. 24.SE - Prob. 29VCCh. 24.SE - Predict the product(s) for each reaction below and...Ch. 24.SE - Predict the product(s) and provide the complete...Ch. 24.SE - Prob. 32MPCh. 24.SE - Predict the product(s) and provide the mechanism...Ch. 24.SE - The diazotization of aniline first involves the...Ch. 24.SE - Prob. 35MPCh. 24.SE - Prob. 36MPCh. 24.SE - Prob. 37MPCh. 24.SE - Prob. 38MPCh. 24.SE - Choline, a component of the phospholipids in cell...Ch. 24.SE - Prob. 40MPCh. 24.SE - -Amino acids can be prepared by the Strecker...Ch. 24.SE - Prob. 42MPCh. 24.SE - Prob. 43MPCh. 24.SE - Prob. 44MPCh. 24.SE - Propose a mechanism for the following reaction:Ch. 24.SE - Prob. 46MPCh. 24.SE - Name the following compounds:Ch. 24.SE - Prob. 48APCh. 24.SE - Prob. 49APCh. 24.SE - Prob. 50APCh. 24.SE - Histamine, whose release in the body triggers...Ch. 24.SE - Prob. 52APCh. 24.SE - Prob. 53APCh. 24.SE - Prob. 54APCh. 24.SE - Prob. 55APCh. 24.SE - Prob. 56APCh. 24.SE - Prob. 57APCh. 24.SE - Prob. 58APCh. 24.SE - Prob. 59APCh. 24.SE - Prob. 60APCh. 24.SE - Show the products from reaction of p-bromoaniline...Ch. 24.SE - Prob. 62APCh. 24.SE - How would you prepare the following compounds from...Ch. 24.SE - Prob. 64APCh. 24.SE - Phenacetin, a substance formerly used in...Ch. 24.SE - Prob. 66APCh. 24.SE - Draw the structure of the amine that produced the...Ch. 24.SE - Fill in the missing reagents a-e in the following...Ch. 24.SE - Prob. 69APCh. 24.SE - Prob. 70APCh. 24.SE - Deduce the structure of the compound with formula...Ch. 24.SE - Prob. 72APCh. 24.SE - Prob. 73APCh. 24.SE - Prob. 74APCh. 24.SE - Prob. 75APCh. 24.SE - Prob. 76APCh. 24.SE - Propose a structure for the product with formula...Ch. 24.SE - Prob. 78APCh. 24.SE - Prob. 79APCh. 24.SE - Prob. 80APCh. 24.SE - Prob. 81APCh. 24.SE - Prob. 82APCh. 24.SE - Prob. 83APCh. 24.SE - Prob. 84AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compound A, MW 86, shows an IR absorption at 1730 cm-1 and a very simple 1H NMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.arrow_forwardPropose a structure with the formula C3H6Br2 that would produce the following H1 NMR spectrum.arrow_forwardPropose a structure for a compound, C10H14, that fits the following 1H NMR data. δ1.30, 9H, singlet δ7.30, 5H, singletarrow_forward
- Treatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forwardTreatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products (M and N) with molecular formula C6H12. The 1H NMR spectra of M and N are given below. Propose structures for M and N, and draw a mechanism to explain their formation.arrow_forwardPropose a structure for each of the following two isomers with formula C6H14 given their 1H-NMR spectra. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (t, 9H), 1.22 (q, 2H) ppmarrow_forward
- A compound gives the following IR spectrum. Upon reaction with sodium borohydride followed by acidification, it forms the product with the 1H NMR spectrum shown below. Identify the starting material and the productarrow_forwardPropose a structural formula for each compound consistent with its 1H-NMR and 13C-NMR spectra.arrow_forwardA compound gives the following IR spectrum. Upon reaction with sodium borohydride followed by acidification, it forms the product with the 1H NMRspectrum shown below. Identify the starting material and the product.arrow_forward
- the following is a 1H NMR spectrum of a C8H10O alcohol, propose a structurearrow_forwardWhat carbonyl compounds are required to prepare a compound with molecular formula C10H10O whose 1H NMR spectrum is shown below?arrow_forwardIdentify the following molecules based on their IR, 13C and 1N NMR spectraarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY