
Interpretation:
In the structures of the given compounds, the carbon atoms that are expected to be labeled with
Concept introduction:
The isotopically labeled carbons in various compounds that are synthesized biologically enriched with
The organic material are dated using radioactive isotope of carbon
Answer to Problem 17P
Solution:
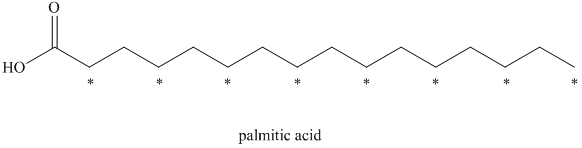
a) There are
b) There are
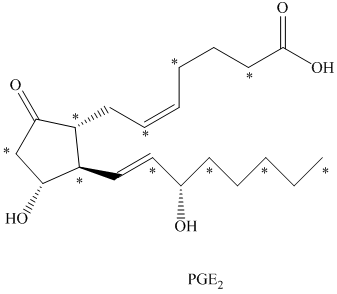
c) There are
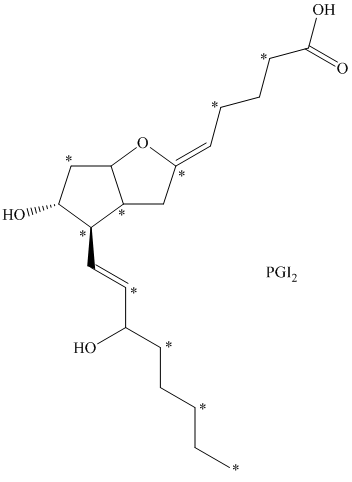
d) There are
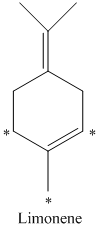
e) There are
Explanation of Solution
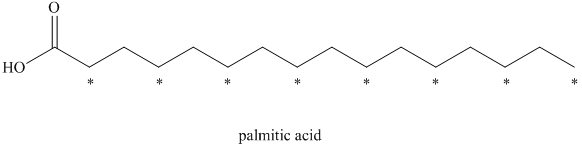
a) The given compound is Palmitic acid.
The isotopically labeled carbon atoms in palmitic acid synthesized from the acetate enriched with
b) The given compound is
The isotopically labeled carbon atoms in
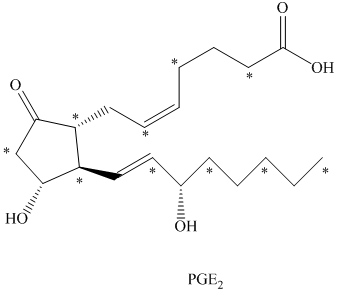
c) The given compound is
The isotopically labeled carbon atoms in
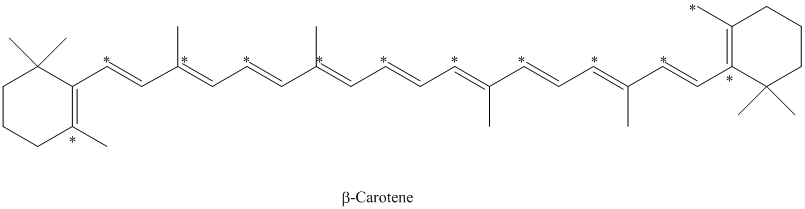
d) The given compound is limonene.
It is synthesized by the geranyl diphosphate that is further synthesized from dimethylallyl phosphate and isopentenyl diphosphate. The isotopically labeled carbon atoms in limonene enriched with
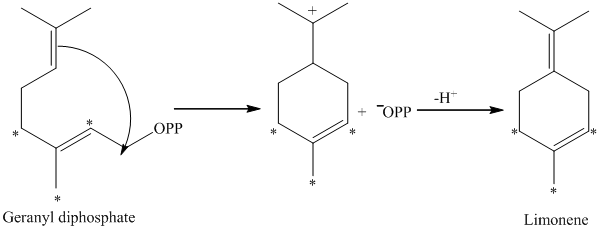
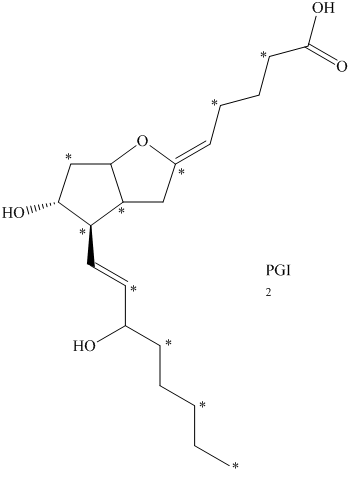
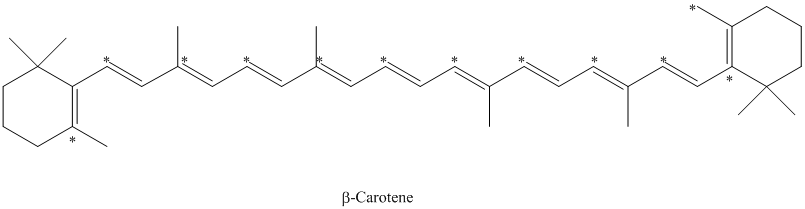
e) The given compound is
The isotopically labeled carbon atoms in
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry
- An organic compound is analysed and found to contain 66.7% carbon, 11.2% hydrogen and 22.1% oxygen by mass. The compound boils at 79.6 C. At 100 C and 0.970atm, the vapour has a density of 2.28g/L. The compound has a carbonyl group and cannot be oxidized to a carboxylic acid. Suggest a structure for the compound.arrow_forwardWhen HOCH2CH2CH2CH2COCH2CH2CH2CH2OH is heated in the presence of an acid catalyst, a reaction occurs. This product has the formula C9H16)2. Provide the structure of this product.arrow_forwardExplain the below ? A phenyl group (C6H5−) is formed by removing one hydrogen frombenzene (C6H6).arrow_forward
- Give a simple chemical test that can differentiate the two compounds from each other. Specify the reagent(s) needed and state what you expect to see. benzene and ethylbenzene 1 – butyne and 2 – butyne 2 – methylpentane and 2 - methylcyclohexenearrow_forwardGive a structure for the unknown that matches all given data. Then give the IUPAC name of the unknown and explain the conclusion (in great detail if possible). The unknown for the data is C6H14O.arrow_forwardDraw a structural formula for compound. Q.) 2,4,6-Trinitrotoluenearrow_forward
- Identify the benzene rings in the structure of fesoterodine?arrow_forward2)Write a balanced chemical equation for each of the following processes, Structural or displayed formulae shouldbe used for all organic substances,a)Making ethanol using ethene as feedstock. Include the formula of the catalyst used.b)The complete combustion of ethanol in oxygen.C)The dehydration of butan-2-ol when passed over hot AI2 O3. Give three equations, one for each of the three possible products,d)The reaction of ethanoic acid with ethanol. Name the catalyst used, the type of reaction andthe products.arrow_forwardPropose a structure that fits the description: a nitrile, C3H5N.arrow_forward
- A compound with formula C7H12O is treated with sodium borohydride in methanol to yield 2,2-dimethylcylopentanol. Write a reaction scheme showing the structures of the reactant, the reagents, and the product. Will the product be optically active? Explain.arrow_forwardHydrocarbon A, C8H12 absorbs 3 equiv. of hydrogen to give B, C8H18 when hydrogenated over a Pd/C catalyst. Treatment of A with aqueous H2SO4 and Hg(II) gives a single ketone, C. Oxidation of A with KMnO4 gives CO2 and the two carboxylic acids D & E shown below. What would be a structure for A?arrow_forwardWhat is the functional group shown in the molecule below? And what are the products if this material is treated with aqueous acid at 30 degrees C for five hours?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





