
ORGANIC CHEMISTRY WILEYPLUS >I<
12th Edition
ISBN: 9781119077213
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 25, Problem 1PP
Interpretation Introduction
Interpretation:
The tautomeric structural forms of Adenine, guanine, cytosine, thymine and uracil are to be written.
Concept Introduction:
A nucleoside is obtained by removing a phosphate group from
The bases obtained from nucleoside are exists in more than one tautomeric forms.
Expert Solution & Answer
Answer to Problem 1PP
Solution:

Explanation of Solution
Given information:
The tautomeric forms of adenine, guanine, cytosine, thymine and uracil is as follows:
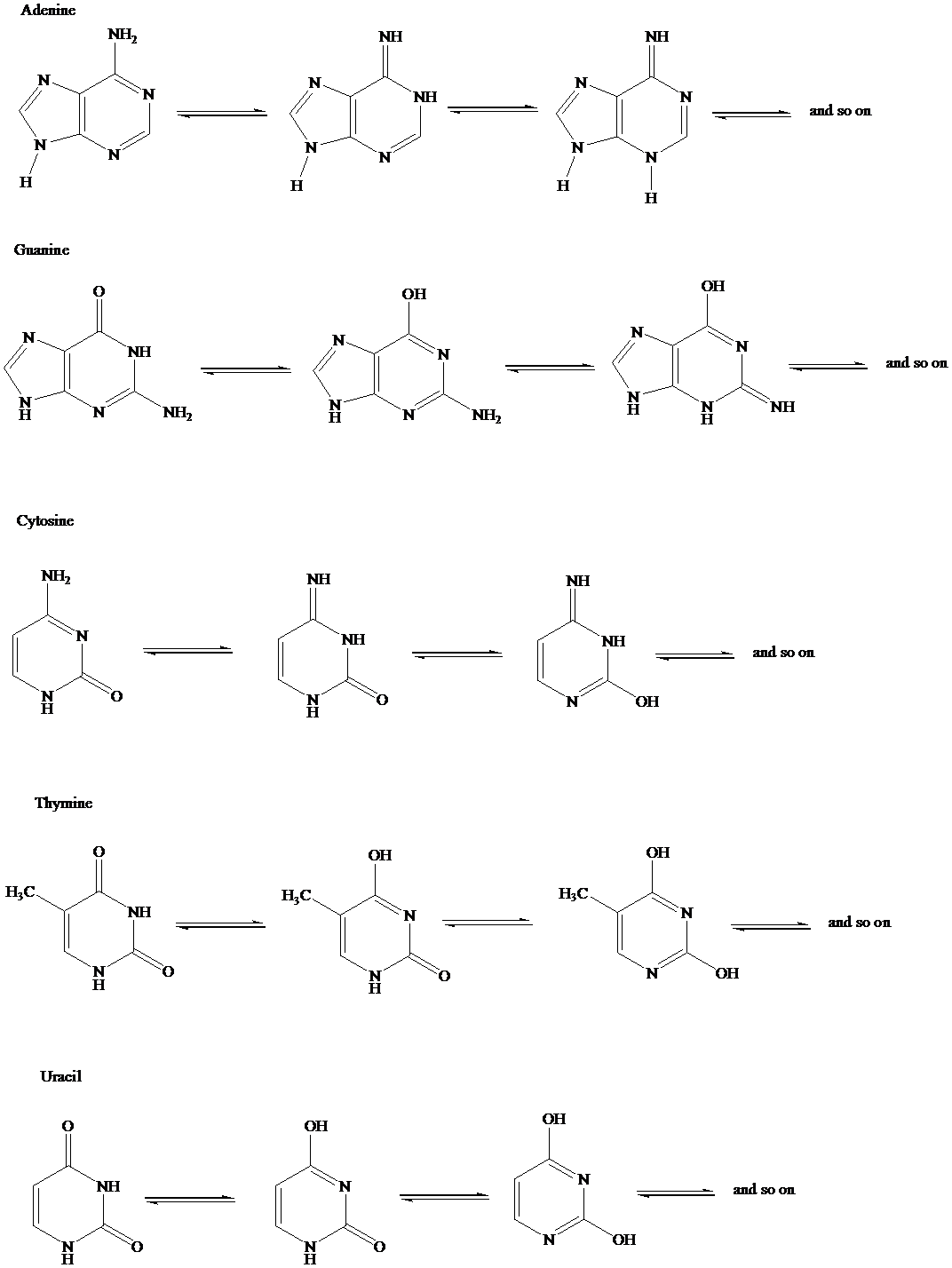
Want to see more full solutions like this?
Subscribe now to access step-by-step solutions to millions of textbook problems written by subject matter experts!
Students have asked these similar questions
In a paragraph form, provide the experimental procedures in the chemical synthesis of protein that contain native serine residues by the reaction of oxazetidine-containing peptides and α-ketoacid.
Suggest a scheme you will follow to synthesis the dipeptide Ala-Gly
Explanation of scheme should be in words with structures
A tripeptide undergoes complete hydrolysis and the resulting mixture contains only phenylalanine and glycine. Draw all possible sequences for the original tripeptide.
Chapter 25 Solutions
ORGANIC CHEMISTRY WILEYPLUS >I<
Ch. 25 - Prob. 1PPCh. 25 - Prob. 2PPCh. 25 - Prob. 3PPCh. 25 - Prob. 4PPCh. 25 - Prob. 5PPCh. 25 - Practice Problem 25.6 (a) There are approximately...Ch. 25 - Prob. 7PPCh. 25 - Prob. 8PPCh. 25 - Prob. 9PPCh. 25 - Practice Problem 25.10
The sense strand of a...
Ch. 25 - Practice Problem 25.11
(a) Using the first codon...Ch. 25 - Write the structure of the RNA dinucleotide GC in...Ch. 25 - 25.13 Write the structure of the DNA dinucleotide...Ch. 25 - The example of a silylHilbertJohnson...Ch. 25 - Prob. 15PCh. 25 - Prob. 16PCh. 25 - Research suggests that expression of certain genes...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structure of leu-enkephalin, a pentapeptide that acts as an analgesic and opiate, and has the following sequence: Tyr–Gly–Gly–Phe–Leu. (The structure of a related peptide, met-enkephalin, appeared in Section 22.6B.)arrow_forwardChemistry Draw this peptide showing correct stereochemistry (hashed/wedged bonds) and please explain thoroughly. N-terminus - Val - Leu - Tyr - Cys - Ala - Gly - C-terminusarrow_forwardUsing alanine as an example, draw possible conformations of alanine in a peptide chain (Ala-Met-Thr) and identify which conformer is favoured.arrow_forward
- Draw the tetrapeptide represented by HLQS at physiological pH (pH=7.4), Include proper stereochemistryarrow_forwardDeduce the sequence of a pentapeptide that contains the amino acids Ala, Glu, Gly, Ser, and Tyr, from the following experimental data. Edman degradation cleaves Gly from the pentapeptide, and carboxypeptidase forms Ala and a tetrapeptide. Treatment of the pentapeptide with chymotrypsin forms a dipeptide and a tripeptide. Partial hydrolysis forms Gly, Ser, and the tripeptide Tyr–Glu–Ala.arrow_forwardDraw the structure of leu-enkephalin, a pentapeptide that acts as an analgesic and opiate, and has the following sequence: Tyr–Gly–Gly–Phe–Leu.arrow_forward
- Alanine has pKa values of 2.34 and 9.69. Therefore, alanine exists predominately as a zwitterion in an aqueous solution with pH > ____ and pH < ____.arrow_forwardWhich of the following is an acceptable name for the peptide sequence below?arrow_forwardThe peptide Proline-Serine-Alanine-Phenylalanine-Glutamine is present at pH 7. Draw the peptide and include stereochemistry.arrow_forward
- A tripeptide on hydrolysis produced glycine, alanine and leucine. The structures of these amino acids are shown below. On reaction with Edman’s reagent, leucine was released as the phenylhydantoin. Treatment of the tripeptide with carboxypeptidase gave glycine. Draw the structure of the tripeptide.arrow_forwardIf a protein were subjected to an acidic solution, which interactions will be disrupted?arrow_forwardAn octapeptide contains the following amino acids: Arg, Glu, His, Ile, Leu, Phe, Tyr, and Val. Carboxypeptidase treatment of the octapeptide forms Phe and a heptapeptide. Treatment of the octapeptide with chymotrypsin forms two tetrapeptides, A and B. Treatment of A with trypsin yields two dipeptides, C and D. Edman degradation cleaves the following amino acids from each peptide: Glu (octapeptide), Glu (A), Ile (B), Glu (C), and Val (D). Partial hydrolysis of tetrapeptide B forms Ile–Leu in addition to other products. Deduce the structure of the octapeptide and fragments A–D.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning