
Concept explainers
(a)
Interpretation:
The structure for each of the given thioesters.
Concept Introduction:
Thioesters are sulfur analogs of esters. The S-alkyl or S-aryl thioester has a carbonyl group and a sulfur-linked alkyl or aryl group. The first part of the name indicates that the alkyl or aryl group has bonded to the sulfur atom. The last part of the name indicates the carboxylic acid stem. In common usage, the “S-“prefix is understood, and so, it is not mentioned.
Answer to Problem 25.1P
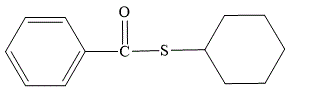
Explanation of Solution
In the given name cyclohexyl thiobenzoate, the cyclohexyl is the alkyl group which is bonded to the sulfur atom. Thiobenzoate consists of a phenyl ring bonded to the sulfur atom through a carbonyl group. The structure for cyclohexyl thiobenzoate is drawn as below:
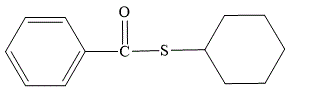
The structure for cyclohexyl thiobenzoate is:
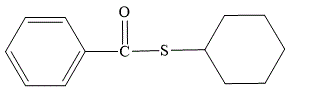
(b)
Interpretation:
The structure for each of the given thioesters.
Concept Introduction:
Thioesters are sulfur analogs of esters. The S-alkyl or S-aryl thioester has a carbonyl group and a sulfur-linked alkyl or aryl group. The first part of the name indicates that the alkyl or aryl group has bonded to the sulfur atom. The last part of the name indicates the carboxylic acid stem. In common usage, the “S-“prefix is understood, and so, it is not mentioned.
Answer to Problem 25.1P
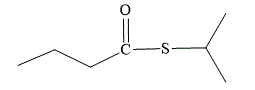
Explanation of Solution
The first part of the name suggests that the isopropyl group must be attached directly to the sulfur atom. The second part of the name suggests that the ester is formed from butanoic acid. In

The structure for S-isopropyl butanethioate is:

(c)
Interpretation:
The structure for each of the given thioesters.
Concept Introduction:
Thioesters are sulfur analogs of esters. The S-alkyl or S-aryl thioester has a carbonyl group and a sulfur-linked alkyl or aryl group. The first part of the name indicates that the alkyl or aryl group has bonded to the sulfur atom. The last part of the name indicates the carboxylic acid stem. In common usage, the “S-“prefix is understood, and so, it is not mentioned.
Answer to Problem 25.1P
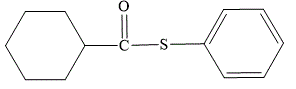
Explanation of Solution
The first part of the name suggests that the phenyl ring must be attached directly to the sulfur atom. The second part of the name suggests that the ester is formed from the carboxylic acid which contains a cyclohexyl ring. In IUPAC nomenclature, “thio-“is inserted before the “ate” suffix in the name of the corresponding oxygen ester.
The structure for S-phenyl cyclohexanecarbothioate is shown as below:

The structure for S-phenyl cyclohexanecarbothioate is:

Want to see more full solutions like this?
Chapter 25 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Quinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure. One step in the synthesis of quinapril involvesreaction of the racemic alkyl bromide A with a single enantiomer of theamino ester B. Given the structure of quinapril, which one of these two products isneeded to synthesize the drug?arrow_forwardAspartame, the sweetener used in the commercial products NutraSweet and Equal, is 200 times sweeter than sucrose. What products will be obtained ifaspartame is hydrolyzed completely in an aqueous solution of HCl?arrow_forwardCompound 1 is an anticoagulant that is extracted from, among other plants, the sweetclover plant, Melilotus Officinalis. However, the compound is moderately toxic and alsofinds use as a rodenticide. Name this compoundarrow_forward
- Give the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)NaOH, H2Oarrow_forwardAlthough ibuprofen is sold as a racemic mixture, only the S enantiomer acts as an analgesic. In the body, however, some of the R enantiomer is converted to the S isomer by tautomerization to an enol and then protonation to regenerate the carbonyl compound. Write a stepwise mechanism for this isomerization.arrow_forwardDraw a line structure clearly showing the stereochemistry of (3S,4S)-4-hydroxy-3,5-dimethylhexanoic acid.arrow_forward
- A chiral amine A having the R conguration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by CH3SCH3, to form CH2 = O and CH3CH2CH2CHO. What are the structures of A and B?arrow_forwardThe cyclic hemiacetal is more stable than the open-chain form, so very little of the open-chain form is present atequilibrium. Will an aqueous solution of glucose reduce Tollens reagent and give a positive Tollens test? Explain.arrow_forwardDraw the structure of each compound.(a) o-nitroanisole (b) 2,4-dimethoxyphenol (c) p-aminobenzoic acid(d) 4-nitroanilinearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


