
Concept explainers
(a)
Interpretation:
The product formed by the reaction of D-ribose with
Concept Introduction:
The reaction of
(a)
Explanation of Solution
The D-ribose is an aldopentose molecule. The carbonyl group present in D-ribose is
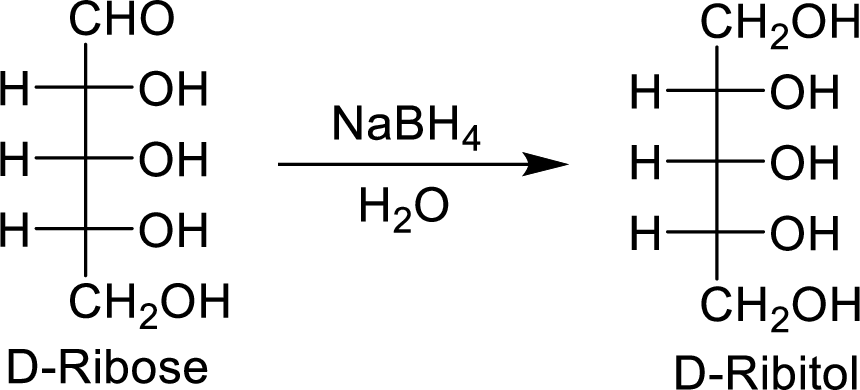
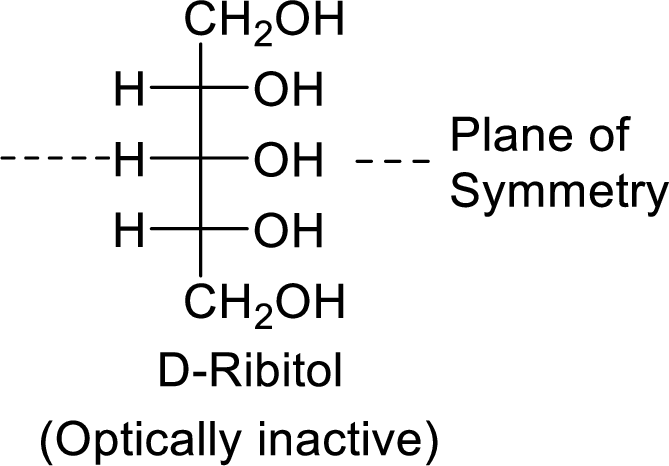
The D-ribitol is a meso compound and shows plane of symmetry. So, it is optically inactive.
(b)
Interpretation:
The product formed by the reaction of D-ribose with
Concept Introduction:
The reagent
(b)
Explanation of Solution
When the D-ribose molecule is made to react with
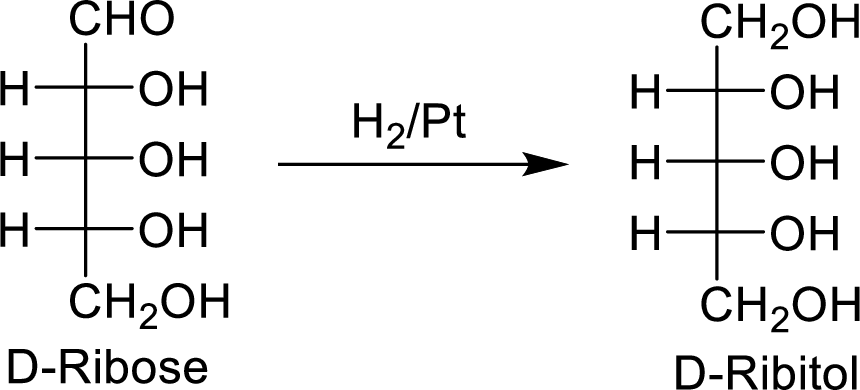
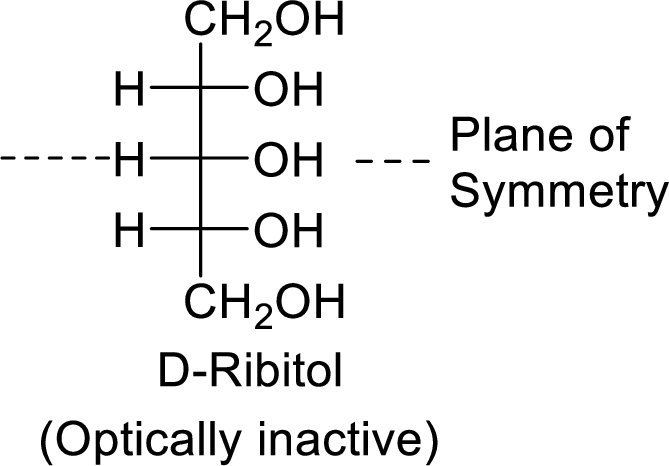
The D-ribitol is a meso compound and shows plane of symmetry. So, it is optically inactive.
(c)
Interpretation:
The product formed by the reaction of D-ribose with warm
Concept Introduction:
The reaction of warm
(c)
Explanation of Solution
The nitric acid (
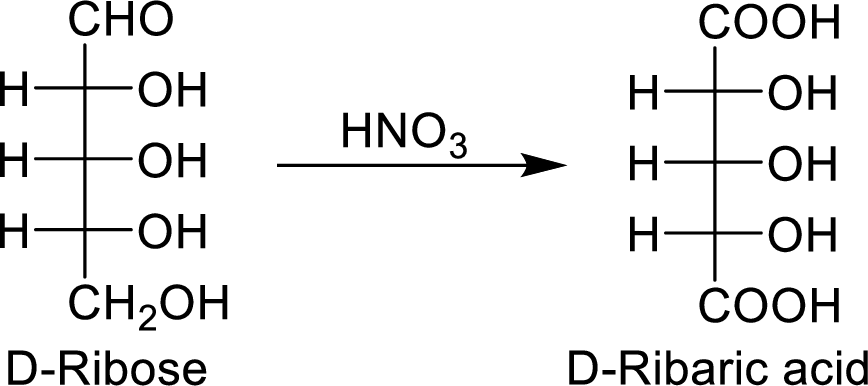
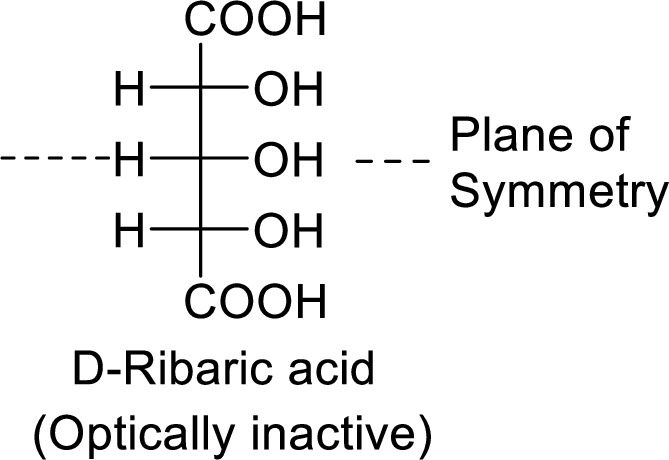
The product formed is a meso compound and shows plane of symmetry. So, it is optically inactive.
(d)
Interpretation:
The product formed by the reaction of D-ribose with
Concept Introduction:
The reaction of
(d)
Explanation of Solution
When the D-ribose reacts with
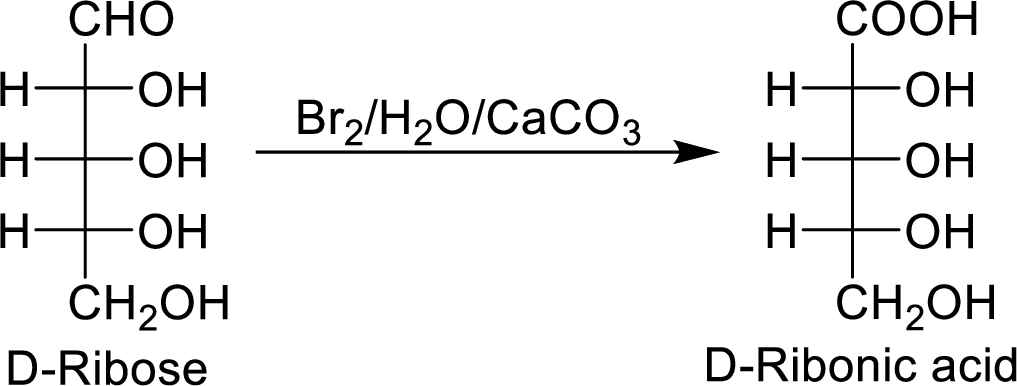
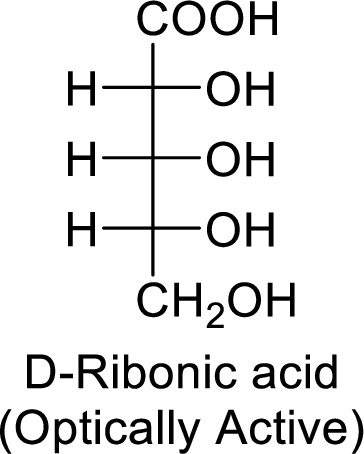
The D-ribonic acid is a chiral molecule and is optically active.
(e)
Interpretation:
The product formed by the reaction of D-ribose with
Concept Introduction:
The reaction of
(e)
Explanation of Solution
The D-ribose molecule consumes
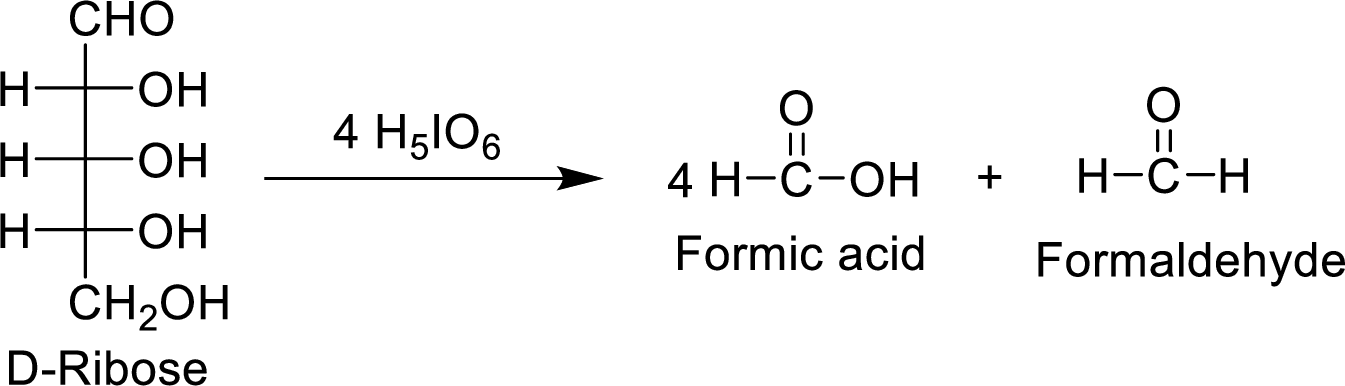
The products formed are achiral and are optically inactive.
(f)
Interpretation:
The product formed by the reaction of D-ribose with aniline (
(f)
Explanation of Solution
The D-galactose molecule reacts with aniline molecule and the aldehyde group of carbohydrate is reacted with the
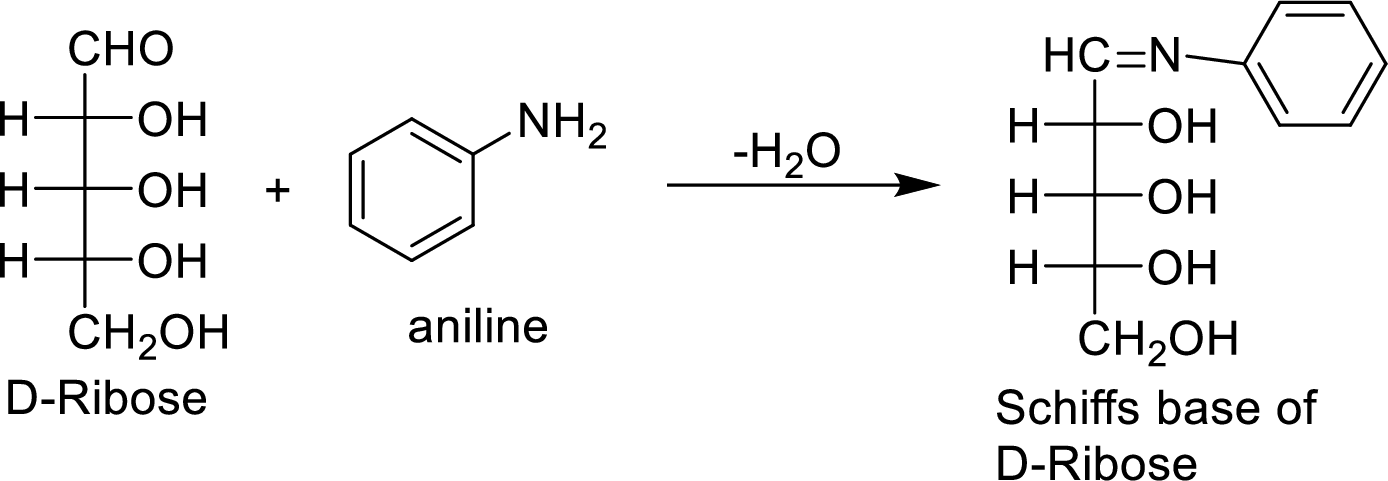
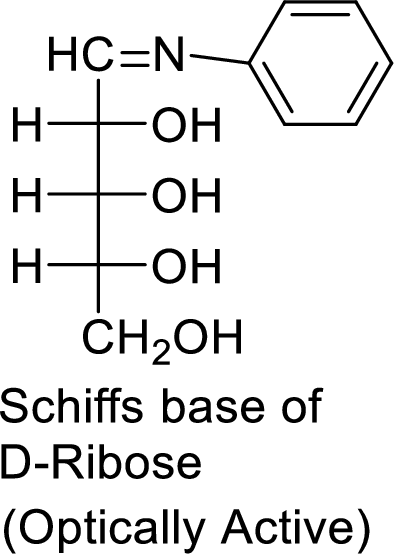
The product formed is chiral and optically active.
Want to see more full solutions like this?
Chapter 25 Solutions
ORGANIC CHEM.(LOOSELEAF)-W/OWL V2 ACCE
- Trehalose and isomaltose are both dimers of glucose. However, they have considerablydifferent reactivities. Concisely explain why these differences are observed.arrow_forwardGlucose is an aldose sugar but it does not react with sodium hydrogensulphite. Give reason.arrow_forwardDeduce the structure of the disaccharide isomaltose from the following data.a. Hydrolysis yields D-glucose exclusively. b. Isomaltose is cleaved with α-glycosidase enzymes. c. Isomaltose is a reducing sugar. d. Methylation with excess CH3I, Ag2O and then hydrolysis with H3O+ forms two products:arrow_forward
- Draw the tetrapeptide represented by HLQS at physiological pH (pH=7.4), Include proper stereochemistryarrow_forwardIllustrate a glycosidic, peptide and a phospho-diester bond.arrow_forwardTwo sugars, A and B, are known to be glucose and galactose, but it is not certain which oneis which. On treatment with nitric acid, A gives an optically inactive aldaric acid, whileB gives an optically active aldaric acid. Which sugar is glucose, and which is galactose?arrow_forward
- An important technique for establishing relative configurations among isomeric aldoses and ketoses is to convert both terminal carbon atoms to the same functional group. This can be done either by selective oxidation or reduction. As a specific example, nitric acid oxidation of d-erythrose gives meso-tartaric acid . Similar oxidation of d-threose gives (2S,3S)-tartaric acid. Given this information and the fact that d-erythrose and d-threose are diastereomers, draw Fischer projections for d-erythrose and d-threose. Check your answers against Table 25.1.arrow_forwardpslease help.with,part.Carrow_forwardRaffinose is a trisaccharide (C18H32O16) isolated from cottonseed meal. Raffinose doesnot reduce Tollens reagent, and it does not mutarotate. Complete hydrolysis of raffinosegives d-glucose, d-fructose, and d-galactose. When raffinose is treated with invertase,the products are d-fructose and a reducing disaccharide called melibiose. Raffinose isunaffected by treatment with a b@galactosidase, but an a@galactosidase hydrolyzes itto d-galactose and sucrose. When raffinose is treated with dimethyl sulfate and basefollowed by hydrolysis, the products are 2,3,4-tri-O-methylglucose, 1,3,4,6-tetraO-methylfructose, and 2,3,4,6-tetra-O-methylgalactose. Determine the completestructures of raffinose and melibiose, and give a systematic name for melibiose.arrow_forward
- d-Altrose is an aldohexose. Ruff degradation of d-altrose gives the same aldopentose asdoes degradation of d-allose, the C3 epimer of glucose. Give the structure of d-altrosearrow_forwardThere are three (3) vials labeled A, B, and C known to contain the following monosaccharides. All three samples reduce Tollens and Fehling. By oxidation with dilute HNO3 an optically active aldaric acid is obtained for sample A and the remaining two give products without optical activity. When the three samples were subjected to an alkaline medium, it was observed that, after a certain time, samples A and C reached the same value of the specific rotation [α]. Select the RIGHT alternative: (a) Sample A is Galactose. (b) Sample B is Alosa. (c) Samples A and C are not related to each other by an epimerization process. (d) Sample C is Talose. (e) Samples B and C are epimers.arrow_forwardFor the following sugar structure, indicate if it is an aldose or ketose, hexose or other “numbered ose”, and if it is D- or L-. Given that it is a furanose, draw its Haworth structure.arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


